Formulas and calculations
Conversions
Knowing common conversions matters when you’re using formulas and doing calculations in pharmacy compounding. Here are key conversions to remember.
Roman numerals
Here are common Roman numerals and what they represent.
Calculations
Concentration
Concentration tells you how much active ingredient is present in a total amount of product.
-
Liquids are often expressed as weight/volume (): the numerator is the drug’s weight, and the denominator is a specific volume of the final mixture (drug + vehicle).
- Example: A suspension labeled contains mg of active drug in each of suspension.
-
Solid and semisolid topical products are often expressed as weight/weight (): the numerator is the drug’s weight, and the denominator is the total weight of the drug plus vehicle.
- Example: Nystatin cream labeled contains units of nystatin in each gram of cream.
-
Some liquid products are expressed as volume/volume (): the numerator is the volume of active ingredient, and the denominator is the total volume of the final liquid.
- Example: means of active drug in of total solution.
Specific gravity is the ratio of the weight of a substance to the weight of an equal volume of water (measured at the same time). If you know the specific gravity, you can convert between a substance’s weight and volume.
Specific gravity
Number of tablets
Use this when the prescribed tablet strength isn’t available.
- Example: A prescription requires of drug A, but you only have tablets.
Number of tablets of drug A tablets.
So, you would dispense four tablets of each to provide a total dose of .
Amount of solution to be given
Use this when you need to calculate the volume of a liquid medication to dispense and you know the stock concentration.
- Example: The stock solution is . You need to dispense . What volume should you dispense?
Using the formula above:
Amount of solution to be given
So, dispense of the stock solution.
Ratios
Ratios are another way to express the strength of a solution or preparation.
In ratio strength, the first number is always , followed by a colon and a second number (for example, ). The units are grams or milliliters, depending on whether the preparation is or .
- A ratio strength can mean:
- g in ml (a liquid preparation), or
- g of drug in g of ointment (a solid/semisolid preparation).
For epinephrine:
- A ratio means gram in ml, which is .
- A ratio means gram in ml, which is .
Percent strength: Percent strength represents the number of grams contained in of product.
Percent weight in volume (w/v): The number of grams in of solution is expressed as . Powdered substances suspended in a liquid vehicle are typically calculated as .
- Example: A solution contains 1 gram of powder in of solution.
Percent volume in volume (v/v): The number of milliliters in of solution is expressed as . A liquid component in a liquid preparation is typically calculated on a basis.
Percent weight in weight (w/w): The number of grams in grams of total dosage form is expressed as . Powdered substances mixed with a solid or semisolid base (for example, ointments) are typically calculated as .
If a solution requires a dilution, the amount of active drug stays constant while the total volume increases.
When two solutions have equal osmotic pressure and salt concentration, they are isotonic. Normal saline contains NaCl in sterile water and is therefore an isotonic crystalloid.
Calculating drug dosage based on body surface area: In some conditions, drug dosage is calculated using body surface area (BSA).
, where is body weight in kg, is body height in meters
Pediatric dosing: Clark’s and Young’s rules are used to calculate drug dosages in the pediatric age group (from birth to about 18 years of age).
According to Clark’s rule, the pediatric dose is obtained by dividing the patient’s weight in pounds by the average standard weight of pounds, then multiplying by the adult dose.
Young’s rule uses the patient’s age to calculate the pediatric dosage.
Fried’s rule also uses age to calculate the pediatric dosage.
Pediatric doses are often stated per body weight, for example, .
Estimating intravenous (IV) flow rates: Intravenous infusions administer fluids directly into the veins. The infusion rate can be calculated in milliliters or in drops.
Drops per minute are abbreviated as gtts/min. The drop factor is the number of drops in . The tubing used determines the drop size:
- Macrodrip tubing may be , , or . Macrodrips are used when rapidly infusing large amounts of fluid.
- Microdrip tubing administers and is typically used in children.
All IV packages clearly label the gtts/mL for that set.
The following formulae can be used to set the IV infusion rate:
Total IV volume/time (hour or minute)
International units (IU): International units are used to denote doses for hormones like insulin, vaccines, vitamins, blood products, etc. They measure the drug’s effect or biological activity, not its weight or mass.
Milliequivalent (mEq): The milliequivalent (mEq) is a unit of measure often used for electrolytes. It indicates the chemical activity (combining power) of an element relative to the activity of of hydrogen.
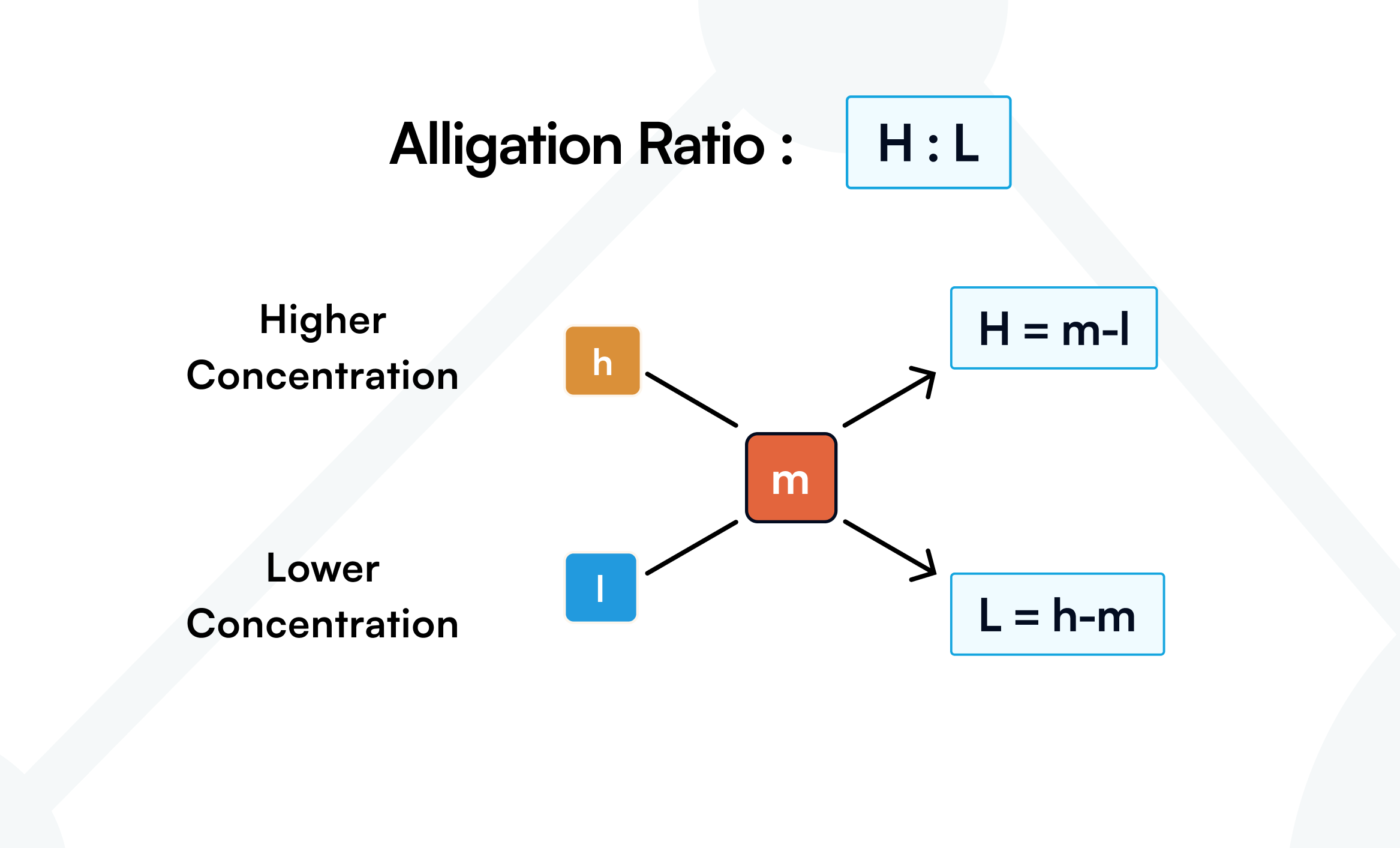
Alligation: Alligation is a method of mixing two liquids or solids of different concentrations to produce a mixture with a desired concentration. The final concentration differs from either starting concentration, so you must mix specific proportions of each ingredient. Those proportions are given by the alligation ratio.
Let “m” be the desired concentration, “l” the lower concentration, and “h” the higher concentration.
Once the alligation ratio is known (for example, ), it means two parts of the higher concentration to three parts of the lower concentration. Next, convert “parts” into actual amounts.
- Example: If you’re compounding total and the ratio is , then:
- Higher concentration:
- Lower concentration:
Example: An order comes in for grams of hydrocortisone cream. You only have and hydrocortisone available. Using the alligation method:
Hence, alligation ratio = .
You need equal parts of and to make . Since you need grams total, mix grams of hydrocortisone with grams of hydrocortisone to get grams of hydrocortisone.
In another example, suppose you get an order for of a solution with a concentration. You have and concentrations available. How will you use the alligation method to calculate the volume of each concentration required?
Alligation ratio =
So you need two parts of the higher concentration () and five parts of the lower concentration (). One way to convert parts into volume is:
Hence, mix of and of to get of .
Dilution: Dilution is a method of preparing a solution of a desired concentration by diluting a concentrated stock solution (typically with water). A common dilution relationship is:
where and are the molarity of the solutions (mol/L or M), and and are the solution volumes. If concentration is given as or , substitute and with those concentrations.
How much water should you add to of a solution to reduce its strength to a solution?
Using the formula above:
Since you already have of solution, the amount of water to add is:
So, add of water to of a solution to obtain a solution.
Make sure you keep units consistent on both sides of the equation (for example, use ml throughout, or convert L to ml as needed).
Aliquot: An aliquot is a dilution method used when only a very small quantity of drug is needed - smaller than the minimum measurable or weighable quantity (MMQ or MWQ) of the available equipment.
- Example: If the MWQ is but you need , you can’t accurately weigh directly.
In that case, weigh the MWQ () and dilute it to a convenient concentration. For example, dilute with of water to make:
Then measure of the solution to obtain of the drug.