Additional information
Prescription label
The prescription label is printed based on the drug information entered into the pharmacy management system. It typically includes the following parts:
- Pharmacy name, address, and contact number
- Prescription number
- Name of the prescriber
- Patient name
- Date filled
- Directions for use
- Medication name, quantity, and number of refills
- Auxiliary labels
- Date after which medication should not be taken
Packaging
The Poison Prevention Packaging Act of 1970 requires that most oral prescription drugs be dispensed in child-resistant packaging. A common example is a child-safety cap that isn’t easily opened by children. Violations of this act can result in penalties, including imprisonment.
Exceptions may be made at the patient’s request in special circumstances (for example, for elderly patients or for patients with hand deformities caused by rheumatoid arthritis).
Variations of pharmaceutical packaging include blister packs, alternative tamper-evident packaging, bottles, vials, ampules, intravenous bags, and calendar packs.
Packaging should protect the drug and help maintain its physical and chemical stability. Light, temperature, and moisture are common causes of drug stability and potency loss. About 500 drugs require some form of light-resistant packaging (for example, doxycycline, zinc sulfate, and hydrocortisone).
Polyvinyl chloride (PVC) and aluminum are common materials used for blister packaging. Many containers are also made of PVC because it’s low cost and durable. However, some medications (for example, insulin, nitroglycerine, and diazepam) react with PVC, so alternate packaging materials such as glass are used.
Lot number and serial number
A lot number is a code that identifies one batch of a product made at the same time. Its purpose is to:
- Identify a specific batch of product
- Track the product’s transaction history, including the manufacturer
- Support product recalls when needed
All products that share a lot number have the same expiration date.
A serial number identifies the lowest sellable unit. The lowest sellable unit is how the manufacturer chooses to sell that product (for example, one bottle of 100 pills).
While a lot number identifies an entire batch, products within the same lot number can have different serial numbers.
Diabetic supplies
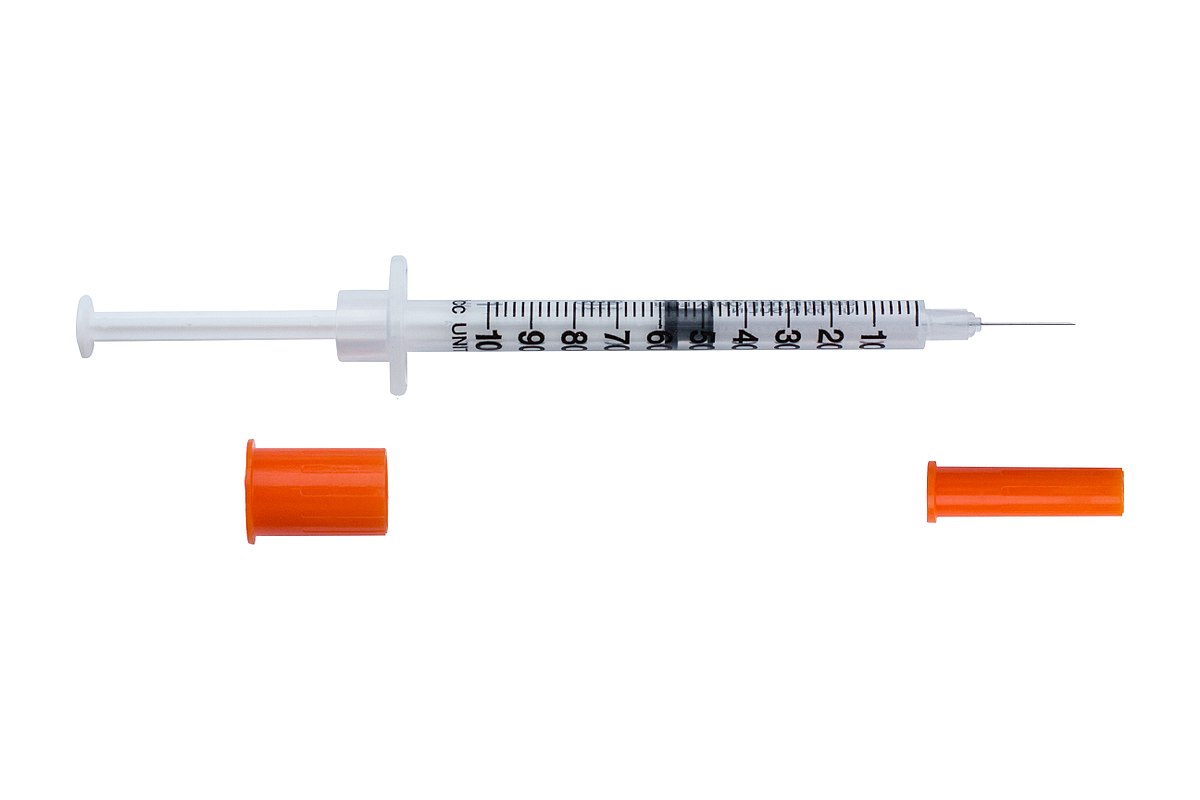
Insulin is often self-administered from an insulin bottle using syringes. Needles attach to syringes and are labeled by length and thickness. Thickness is described using “G” for gauge.
The key relationship to remember is:
- The thinner the needle, the higher its gauge
For example, a 31-gauge needle is thinner than a 28-gauge needle.
Syringes and insulin pens deliver insulin through a needle. Some pens use cartridges that are inserted into the pen. Others are pre-filled and discarded after all the insulin is used. With a pen, the insulin dose is dialed, and the insulin is injected through a needle.
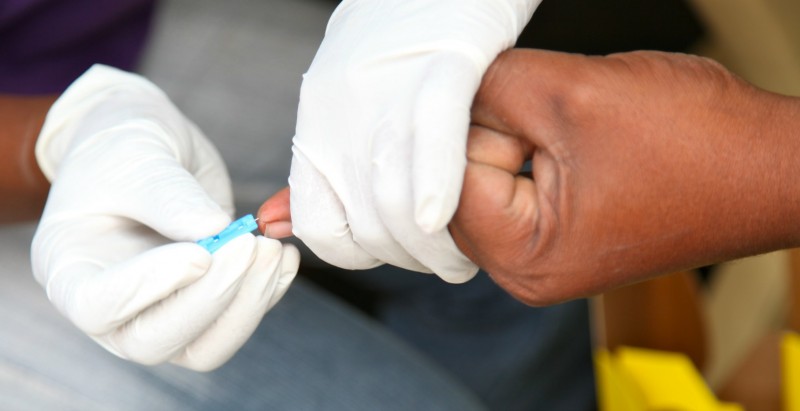
Lancet devices are used to check blood glucose levels by finger prick. They consist of a small, sharp, needle-like lancet and a lancet holder. OneTouch and Freestyle are common brands for lancets.
A glucometer measures blood glucose levels. It uses a lancet device and a test strip to measure glucose.
Insulin pumps are small wearable devices surgically placed under the skin of the stomach or upper arm. They deliver automatically adjusted insulin doses after measuring blood glucose levels. Some insulin pumps use adhesive skin patches to deliver insulin.
Continuous glucose monitoring devices (CGMs) record blood glucose levels around the clock. Dexcom, FreeStyle Libre, and Medtronic are common CGM devices.
iLet® Bionic Pancreas System, Medtronic MiniMed™, Omnipod®, and Tandem® t: slim are insulin pump systems available in the United States.
Procedures for identifying and returning dispensable, non-dispensable, and expired medications and supplies
Medication stock needs to be maintained and reviewed consistently to support efficient dispensing. Inventory can be affected by changing demand, supply chain issues, expired drugs, drug overstocking, and drug returns.
Prescription medication must not be dispensed after the manufacturer’s container expiration date. An internal inventory inspection must be performed at least once every month to verify expiration dates and remove expired drugs.
Unless otherwise stated by the manufacturer or by a stricter facility policy, open medications generally expire on the printed expiration date or one year after the open date (whichever is sooner).
Drugs that are close to expiring are removed from pharmacy shelves and are typically returned to wholesalers. Reverse distribution is when a third party takes the pharmacy’s unsalable, expired drug products and sends them back to manufacturers and/or wholesalers for credit, or disposes of them.
Return credits are a benefit offered by many pharmaceutical manufacturers and distributors. These credits allow pharmacies to return expired or unsold medications for a credit or refund, which can help offset the costs of purchasing and storing these drugs.
Medications not picked up or used 14 days from the fill date are returned to stock (RTS). The patient is sent reminders using their preferred contact method before the medication is returned to stock.
Before restocking, the medication contents are inspected and identified to confirm label accuracy. All return-to-stock medications must have the drug, strength, dosage form, NDC number, manufacturer, and expiration date clearly identified on the label or product packaging. All patient identifiers must be removed before returning the medication to the shelf.