Covalent bonds
Covalent bonds form when two atoms share electrons, which happens when their electron orbitals overlap. By sharing electrons, each atom can fill its valence shell and become more stable.
In covalent bonding, the first bond formed between two atoms is a sigma () bond. A sigma bond comes from direct orbital overlap along the internuclear axis (the line connecting the two nuclei).
If two atoms form multiple bonds, the additional bonds are pi () bonds:
- A double bond = 1 sigma bond + 1 pi bond
- A triple bond = 1 sigma bond + 2 pi bonds
To explain molecular shapes, atoms can form hybrid orbitals by mixing standard atomic orbitals. Common types include:
- hybridization: one s orbital + three p orbitals. This produces four equivalent hybrid orbitals arranged in a tetrahedral geometry, typically forming only single bonds.
- hybridization: one s orbital + two p orbitals. This produces three hybrid orbitals in a trigonal planar arrangement; molecules with centers often have a double bond.
- hybridization: one s orbital + one p orbital. This produces two hybrid orbitals in a linear arrangement, characteristic of triple bonds.
The Valence Shell Electron Pair Repulsion (VSEPR) theory further explains molecular geometry. It predicts that electron pairs around a central atom arrange themselves as far apart as possible to minimize repulsion. This helps explain the three-dimensional shapes of molecules like (trigonal pyramidal), (bent), and (linear).
Lewis electron dot formulas
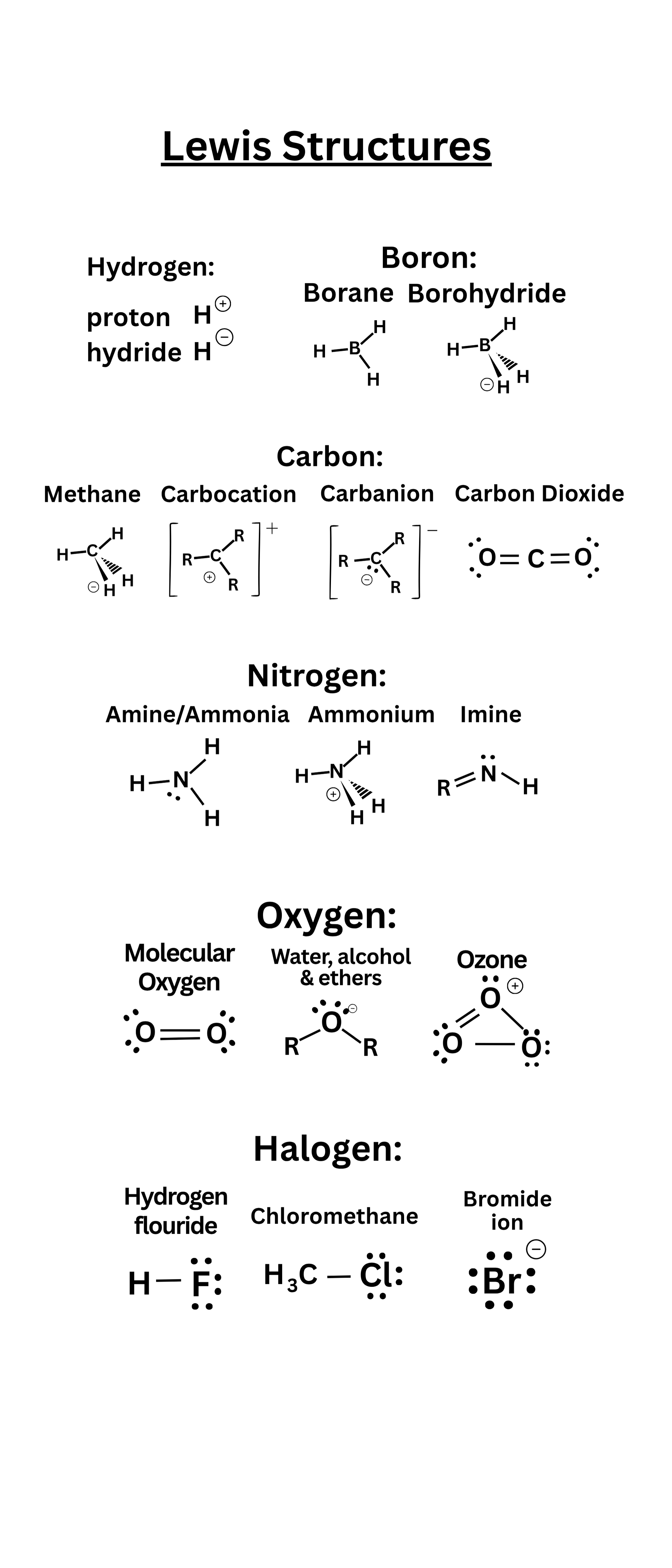
Lewis electron dot formulas are diagrams that show valence electrons as dots around atoms and covalent bonds as lines.
- Each dot represents 1 electron.
- Each line represents 1 shared pair of electrons (2 electrons total).
In these structures:
- A lone pair is shown as two adjacent dots on a single atom.
- Most atoms tend to follow the octet rule (8 electrons around them), though there are notable exceptions.
- A formal charge on an atom is found by comparing the atom’s number of valence electrons in a structure to its valence electrons as a free atom.
- Radicals are species with an unpaired electron.
General features:
- Each dot = 1 electron.
- Each line = 1 bond (2 electrons).
- A lone pair = two dots on a single atom.
- Octet rule: Most main-group elements seek 8 electrons in their valence shell.
- Shared electrons in a bond count toward the octet of both atoms involved.
Exceptions
- The boron column (Group 13) often forms only 3 bonds (6 electrons total around ).
- Heavier elements in the third period or below (e.g., P, S) can expand beyond 8 electrons (e.g., 10 electrons for P in , 12 for S in )
- Radicals with an odd total number of electrons must have at least one unpaired electron.
Rules of thumb for specific elements
-
Carbon (): Typically 4 total bonds and no lone pairs. Examples include (methane) or (carbon dioxide).
-
Oxygen ():
- Neutral : 2 bonds, 2 lone pairs. E.g., .
- : 1 bond, 3 lone pairs, overall −1 charge.
- : 3 bonds, 1 lone pair, overall +1 charge.
-
Nitrogen ():
- Neutral : 3 bonds, 1 lone pair. E.g., (ammonia).
- N⁺: 4 bonds, 0 lone pairs, overall +1 charge. E.g., (ammonium).
-
Halogens (, , , ): 1 bond, 3 lone pairs in neutral forms. Example: in .
-
Hydrogen (): 1 bond, 0 lone pairs. Hydrogen is an exception to the octet rule because it only requires 2 electrons (a “duet”).
-
Carbocation (): 3 bonds, no lone pairs, overall +1 charge.
-
Carbanion (): 3 bonds, 1 lone pair, overall −1 charge.
-
Boron (): 3 bonds, 0 lone pairs (6 electrons total around B). E.g., (borane).
Examples of Lewis structures:
Similarity across a group (column)
Elements in the same group often show similar Lewis structure patterns because they have the same number of valence electrons. For example, sulfur often appears in bonding patterns similar to oxygen. One key difference is that sulfur can expand its valence shell (for example, in with 12 electrons around S). So, in many basic diagrams, you can often substitute S for O, while keeping sulfur’s ability to exceed the octet in mind.
Lewis acids and bases
In Lewis theory:
- A Lewis acid is an electron pair acceptor.
- A Lewis base is an electron pair donor.
Unlike the Brønsted-Lowry definition (which focuses on protons), the Lewis definition focuses on electron pair movement. When a Lewis base donates an electron pair to a Lewis acid, a coordinate covalent bond forms, helping satisfy the acid’s electron deficiency. This idea explains many reactions, including metal-ligand complex formation, where an electron-deficient metal ion accepts electron pairs from a ligand.
Resonance structures
In both ions and molecules, resonance describes situations where electrons aren’t confined to a single bond or atom. Instead, they’re delocalized across multiple atoms. When drawing resonance structures, this is why electrons are shown as being “pushed” to indicate how electron density can be redistributed.
- In ions, delocalization helps distribute charge.
- In molecules, delocalization is especially common in aromatic rings and conjugated double-bond systems.
Resonance structures occur when a molecule can be represented by more than one acceptable Lewis structure. The molecule does not rapidly switch between these drawings; instead, it exists as a resonance hybrid that reflects features of all valid forms, with more stable forms contributing more. For example, if one resonance form shows a single bond and another shows a double bond, the actual bond length is intermediate between a typical single and a typical double bond.
In stable resonance forms, the octet rule is satisfied for each atom (except for known exceptions like boron and hydrogen), and formal charges are minimized or arranged so that like charges are separated while opposite charges are positioned close together.
Partial ionic character
Partial ionic character occurs when a covalent bond forms between atoms with different electronegativities. The more electronegative atom pulls electron density closer, becoming partially negative, while the other atom becomes partially positive.
This unequal charge distribution creates a bond dipole. If the molecule’s overall shape is asymmetrical, the bond dipoles don’t cancel, and the molecule has a net dipole moment (it’s polar). The size of the dipole moment depends on:
- how much charge is separated, and
- the distance between the partial charges.
For example, water () has a bent structure, so its bond dipoles add to give a net dipole. In contrast, has polar bonds, but its geometry is symmetrical, so the individual dipoles cancel and the molecule is non-polar.
σ and π bonds A (sigma) bond forms by direct, head-on overlap of orbitals along the internuclear axis, creating a cylindrically symmetrical region of electron density between two atoms. A (pi) bond forms by side-by-side overlap of p orbitals, creating electron density above and below (or in some cases in front of and behind) the internuclear axis. Together, these ideas explain how single, double, and triple bonds form in molecules containing H, C, N, O, F, S, P, Si, and Cl.
| Element | Valence e- | Usual
bonds | Typically found in |
|---------|------------|---------------|--------------------| | H | 1 | 1 | Hydrocarbon (alkane, alkene, alkyne), hydride. All organic compounds contain hydrogen. | | C | 4 | 4 | Alkane, alkene, alkyne, aromatic rings. All organic compounds contain carbon. | | N | 5 | 3 | Amine, amide, imine, hydrazone, oxime, nitro compound, diazo compound, nitrile/cyanide, azide | | O | 6 | 2 | Alcohol, ether, aldehyde, ketone, carboxylic acid, acyl halide, anhydride, amide, ester, ozone | | F | 7 | 1 | Fluoride | | S | 6 | 2 or 6 | Thiol, sulfide, sulfate, sulfite | | P | 5 | 3 or 5 | Phosphorous compound, phosphate, phosphite | | Si | 4 | 4 | Silane, silicon dioxide | | Cl | 7 | 1 | Chloride, hypochlorite, chlorite, chlorate, perchlorate |