Multiple bonding and stereochemistry
Multiple bonding
Multiple bonding happens when atoms share more than one pair of electrons, forming a double bond (two shared pairs) or a triple bond (three shared pairs). Sharing extra electron pairs pulls the atoms closer together, which:
- Shortens the bond length
- Increases the bond energy, making the bond stronger
Multiple bonds also make parts of a molecule more rigid. Unlike single bonds, double and triple bonds restrict free rotation. Even bonds with partial double-bond character - such as the peptide bond - limit rotation and help lock a molecule into a more fixed geometry.
Stereochemistry of covalently bonded molecules
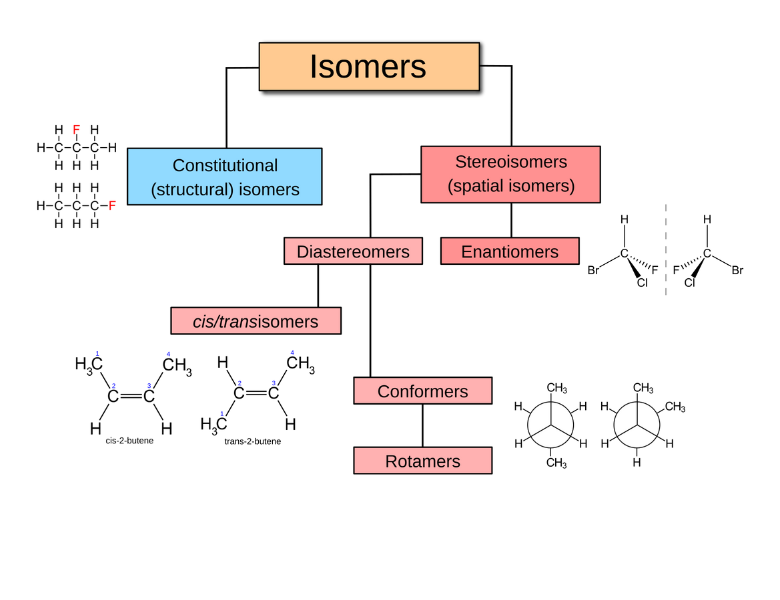
Isomers are compounds with the same molecular formula but different structural arrangements. In other words, they contain the same kinds and numbers of atoms, but those atoms are connected differently or arranged differently in space.
Structural isomers
- Positional isomers have the same functional groups, but those groups appear at different positions in the molecule.
- Constitutional isomers (or structural isomers) have the same formula but differ in how the atoms are connected.
- Functional isomers have the same molecular formula but different functional groups.
Geometric isomers
Geometric isomers have the same molecular formula and the same connectivity, but they differ in how groups are arranged around a double bond:
- When each carbon of the double bond has the same two groups attached, the labels cis (same side) and trans (opposite sides) are used.
- If different groups are attached to each side of the double bond, the labels Z (higher-priority groups on the same side) and E (higher-priority groups on opposite sides) are used, based on the Cahn-Ingold-Prelog rules.
Stereoisomers
Stereoisomers have the same molecular formula and the same atom connectivity, but they differ in the three-dimensional arrangement of atoms:
- Enantiomers are non-superimposable mirror images. Every chiral center (an atom bonded to four distinct groups) is inverted between the two molecules.
- Diastereomers arise when molecules with multiple chiral centers differ at some, but not all, of those centers. In cyclic compounds, cis/trans notation is often used: cis means substituents are on the same side of the ring, and trans means they are on opposite sides.
- Meso compounds contain chiral centers but are overall achiral and optically inactive, which reduces the total number of possible stereoisomers.
Conformational isomers
Conformers are different spatial arrangements of the same molecule that result from rotation around single bonds:
- In eclipsed conformations, groups line up closely. This increases torsional strain and lowers stability. Some eclipsed forms, such as the syn-periplanar conformation, are especially strained.
- Staggered conformations reduce torsional strain:
- Gauche conformations place groups about 60° apart and have moderate stability.
- Anti conformations place groups 180° apart and are the most stable.
- In cyclic molecules (such as hexoses), the chair conformation is favored because it maintains a largely staggered arrangement. The boat conformation is less stable due to increased eclipsing interactions.
- Steric interactions also affect stability:
- Axial positions can create clashes between bulky groups.
- Equatorial positions let substituents extend outward, reducing repulsion and increasing stability.
These stereochemical differences matter because a molecule’s three-dimensional shape strongly influences its physical and chemical properties.
Polarization of light and specific rotation
Light is an electromagnetic wave made of oscillating electric and magnetic fields. These fields are in phase, perpendicular to each other, and perpendicular to the direction the light travels.
In ordinary (unpolarized) light, the electric field vibrates in many directions around the propagation axis (across a full 360°). When light is polarized, the electric field is restricted to a single plane.
Specific rotation describes how a chiral molecule - when present as a single enantiomer - rotates the plane of polarized light. This optical activity can rotate light:
- To the left: negative, l, or levorotatory
- To the right: positive, d, or dextrorotatory
These rotation labels do not correspond to R/S configurations. Also, the lower-case d and l are different from the upper-case D and L used to describe absolute configurations in sugars.