Separation and purification methods
Extraction: distribution of solute between two immiscible solvents
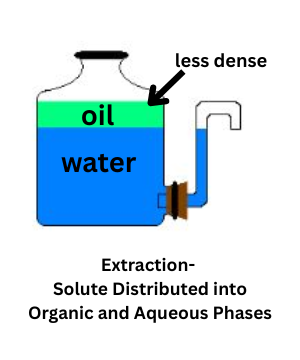
Extraction is a separation method that distributes a solute between two immiscible liquids (liquids that don’t mix). It’s typically done in a separatory funnel and works because the solute has different solubilities in the two layers: the organic phase and the aqueous phase.
The organic phase is a nonpolar solvent, so nonpolar solutes tend to dissolve there. The aqueous phase is usually water, which dissolves ionic and polar compounds more readily. The relative density of the two solvents determines which layer sits on top. Water is denser than many organic solvents, but some organic solvents (such as chloroform) are denser than water and will form the bottom layer.
Distillation
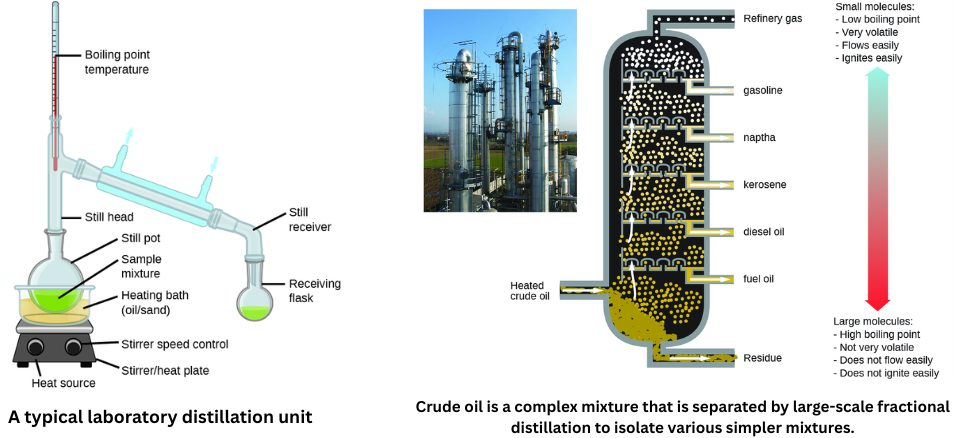
Distillation is a separation technique that uses differences in boiling point and vapor pressure between liquid components. In a mixture of two volatile liquids, the component with the lower boiling point vaporizes more readily. According to Raoult’s law, the vapor phase becomes enriched in the more volatile component. By evaporating, condensing, and collecting that vapor, you can separate it from the less volatile component.
Distillation is widely used in laboratory and industrial settings, including petroleum refining, isolating fermentation products, and purifying water.
In oil refineries, fractional distillation is used. Crude oil is heated at the base of a tall fractionating column, causing many components to vaporize. As the vapors rise, they cool in different regions of the column, condense at different heights, and are collected as distinct fractions for various uses (e.g., diesel, kerosene, gasoline).
There are three types of distillation:
-
Simple distillation uses a standard column and is effective when the boiling point difference between the liquids is large.
-
Vacuum distillation operates under reduced pressure, which lowers the boiling points of all components and helps prevent decomposition that could occur at higher temperatures.
-
Fractional distillation uses a fractionating column to separate liquids with only slight differences in boiling points. Unlike simple distillation, this method relies on repeated vaporization and condensation cycles within the column. As the mixture is heated, the more volatile liquid becomes increasingly concentrated in the vapor phase, allowing more precise separation even when the boiling points are very close.
Chromatography: basic principles involved in separation process
Chromatography separates mixture components based on how they distribute between two phases:
- the mobile phase, which moves through the system
- the stationary phase, which stays in place
An analyte (substrate) repeatedly partitions between these phases. Components that interact more strongly with the stationary phase move more slowly, while components that prefer the mobile phase move more quickly. This difference in movement produces separation.
Gas-liquid chromatography (also called gas chromatography) is used when the analyte can be converted into a gas. Here, the mobile phase is a gas, and the stationary phase is a liquid coating on the inside of a column. As the sample travels through the column, each component equilibrates between the gas and the liquid coating. Components with stronger attraction to the stationary phase spend more time in that phase and therefore emerge from the column later.
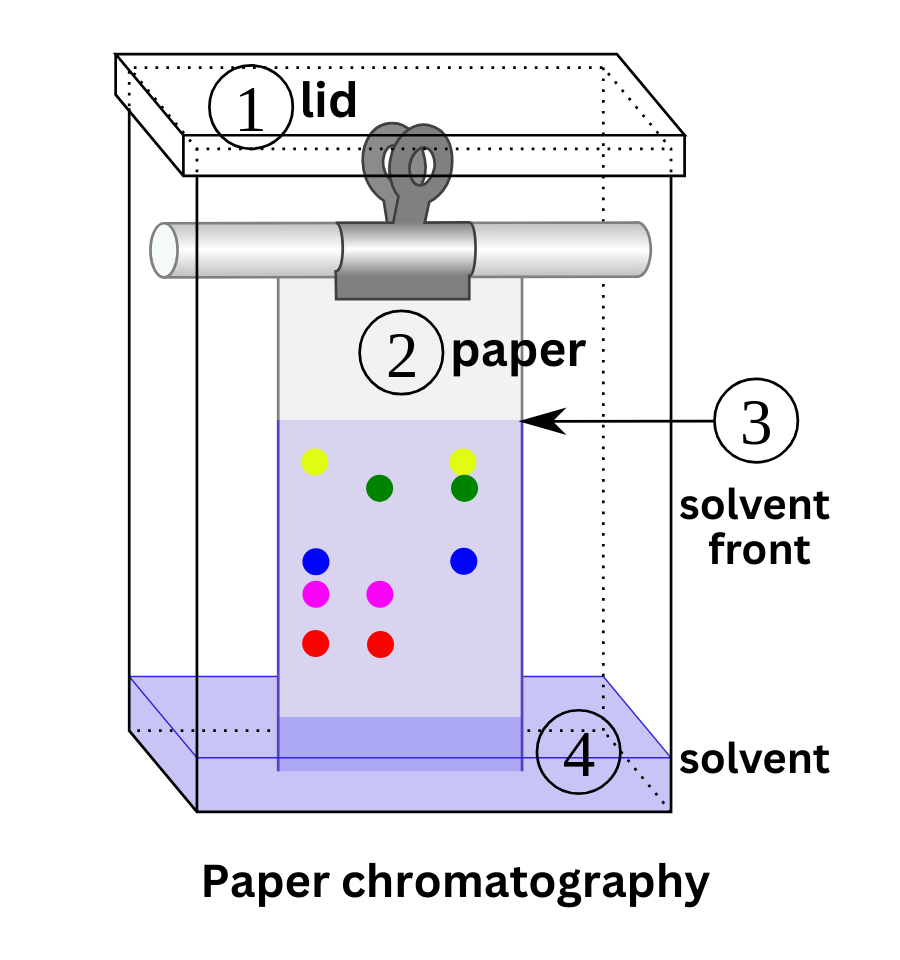
Paper chromatography has traditionally been used to separate pigments in dyes. In this method, the solvent is the mobile phase, and the paper is the stationary phase. As the solvent migrates through the paper, it carries the pigments with it. Pigments with higher affinity for the paper stay closer to the starting spot, while pigments that are more soluble in the solvent travel farther.
Separation is quantified using the Rf value, defined as the ratio of:
- the distance traveled by the pigment
- to the distance traveled by the solvent front
An Rf value of 0 means the pigment hasn’t moved, while an Rf value of 1 means it has moved as far as the solvent front.
Thin-layer chromatography is a more advanced variant that follows the same principles as paper chromatography. Instead of paper, it uses a plate coated with a specific stationary phase. The Rf value is calculated the same way, allowing you to compare how components move under controlled conditions.
Separation and purification of peptides and proteins
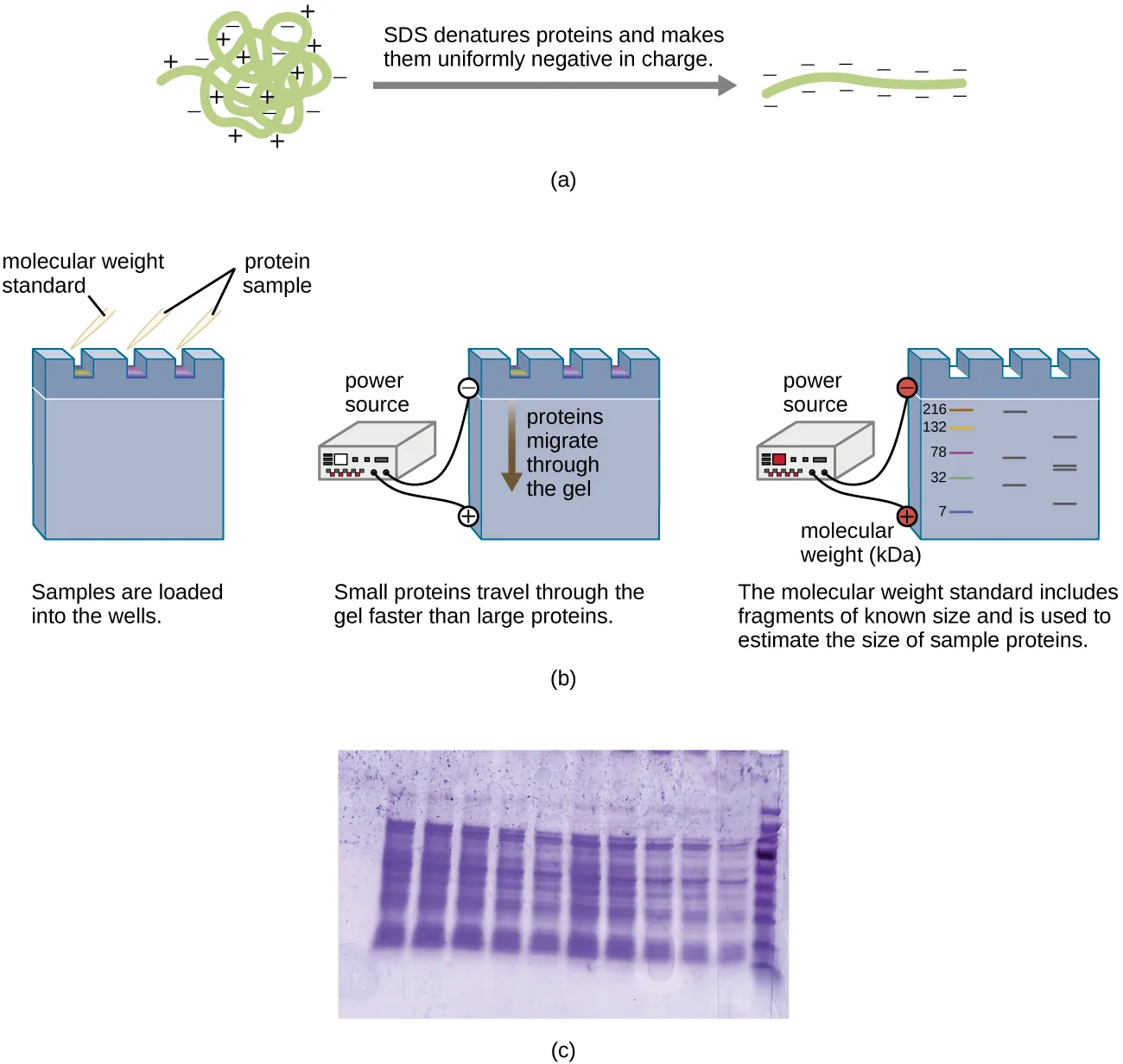
Peptides and proteins can be isolated and purified based on size or charge, and then structurally characterized using several techniques. One widely used method is gel electrophoresis. Proteins or peptides are loaded into a slot near the negatively charged electrode on a porous matrix (commonly polyacrylamide gel). When an electric field is applied, the molecules migrate toward the positive electrode. Smaller or more compact proteins move faster through the pores. To estimate size, a molecular weight standard is typically run alongside the samples for comparison.
Analysis
Proteins can be analyzed in their native form (native-PAGE) or under denaturing conditions (SDS-PAGE).
In native-PAGE, proteins keep their tertiary structure and separate based on both size and shape. Larger or more globular proteins move more slowly.
In SDS-PAGE, the detergent SDS unfolds proteins so they separate primarily by size. Any remaining disulfide bonds can be broken with a reducing agent, helping ensure the polypeptides are fully unfolded.
Another powerful technique is size-exclusion chromatography. Molecules in solution pass through a column packed with porous beads. Smaller molecules can enter the pores, which slows their progress through the column. Larger molecules bypass many pores and elute more quickly. As a result, smaller proteins or peptides have longer retention times, while larger ones exit the column sooner.
Lastly, ion exchange chromatography separates proteins by charge using a charged stationary phase.
- In cation-exchange chromatography, the stationary phase is negatively charged and binds positively charged species (cations).
- In anion-exchange chromatography, the stationary phase is positively charged and binds negatively charged species (anions).
After binding, proteins can be released (eluted) by increasing ionic strength or by adjusting pH to change the protein’s net charge.
Once peptides and proteins are separated, a variety of analytical methods can further identify and characterize them:
- Mass spectrometry determines molecular composition by ionizing the sample and breaking it into fragments, which are then detected based on their mass-to-charge ratio. This approach provides both qualitative and quantitative information about the protein or peptide of interest.
- X-ray crystallography reveals the detailed three-dimensional arrangement of atoms within a protein crystal. By directing X-rays at the crystallized sample and measuring the resulting diffraction patterns, researchers can deduce the protein’s structure at atomic resolution.
- Nuclear magnetic resonance (NMR) spectroscopy uses the magnetic properties of specific atoms to determine a protein’s composition and spatial configuration in solution. However, NMR analysis of proteins is more intricate than that of smaller organic molecules due to the greater complexity of protein structures and the higher number of atomic interactions involved.
Racemic mixtures
Racemic mixtures contain equal proportions of two enantiomers, which are non-superimposable mirror images of each other despite having identical connectivity. Each enantiomer rotates plane-polarized light in the opposite direction but by the same magnitude. In a 50:50 mixture, these rotations cancel, so the solution is optically inactive. Separating the enantiomers from one another is called resolving the racemic mixture.