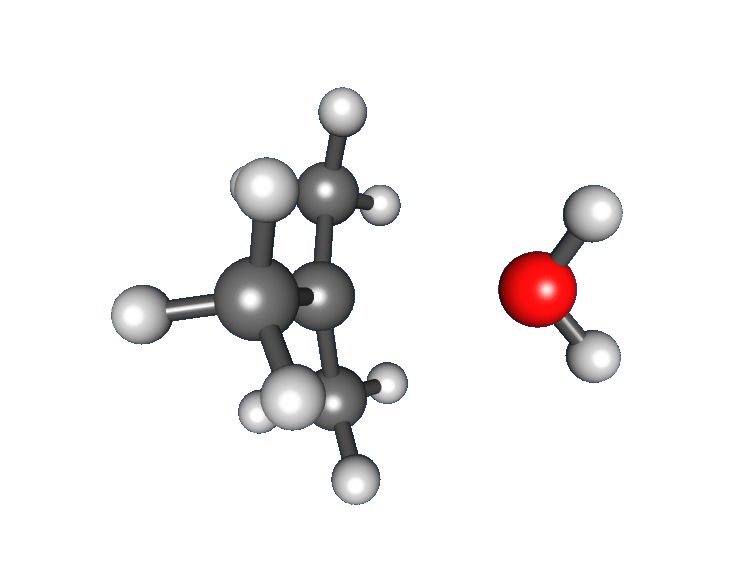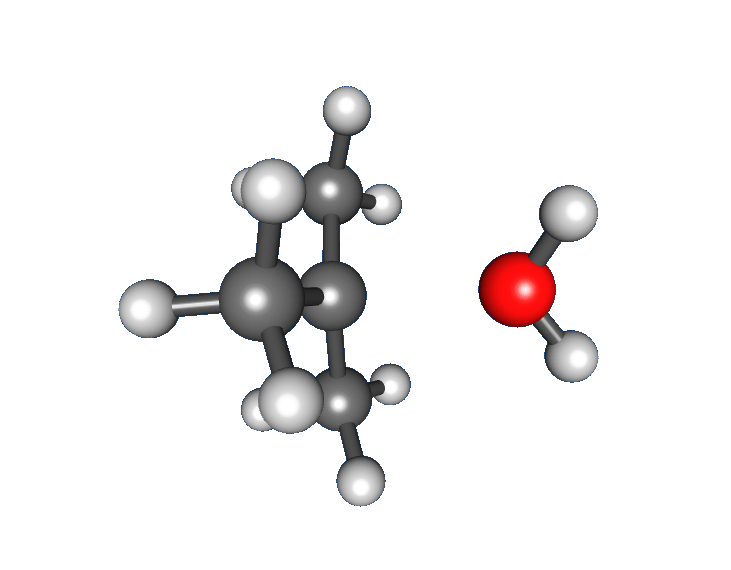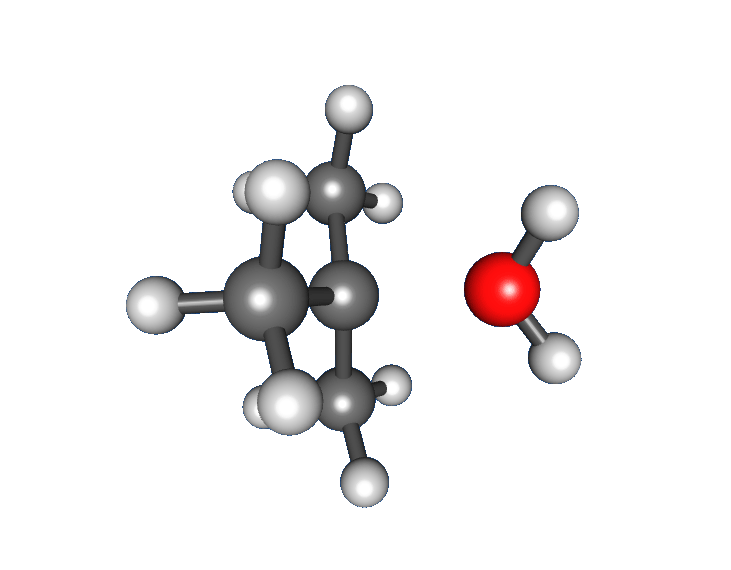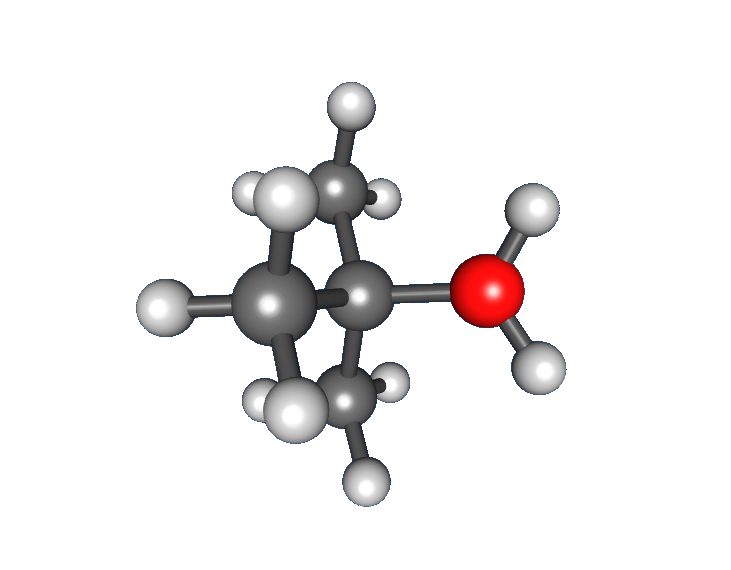Alcohols and carboxylic acids
Alcohols
Alcohols contain a hydroxyl group (). This functional group changes both physical properties (like boiling point and solubility) and chemical behavior compared with hydrocarbons. In synthesis, alcohols often serve as useful intermediates because the group can be oxidized or replaced in substitution reactions.
Nomenclature
Alcohols are commonly named with the suffix “-ol” (e.g., methanol, ethanol). In more complex molecules, “hydroxy” may be used as a prefix to show an substituent. When you name an alcohol, number the longest carbon chain so the hydroxyl group gets the lowest possible number.
Physical properties
Alcohols form hydrogen bonds, which gives them much higher boiling points than analogous hydrocarbons. They dissolve readily in water as long as the carbon chain is relatively short; solubility decreases as the nonpolar carbon chain gets longer. In IR spectroscopy, the stretch appears as a broad peak near 3300 .
Important reactions
-
Substitution reactions: SN1 or SN2
-
- SN1 proceeds through a carbocation intermediate, so it’s favored by tertiary centers and protic solvents.
- SN2 is a one-step (concerted) mechanism, typical of primary centers under polar aprotic conditions.
- Both pathways require a good leaving group, so the is often converted into a sulfonate ester or a halide first.
-
-
Oxidation
- Primary alcohols oxidize to carboxylic acids under strong oxidizers (), or to aldehydes with milder reagents like PCC.
- Secondary alcohols oxidize to ketones.
- Tertiary alcohols generally do not oxidize because they lack α-hydrogens.
- Pinacol rearrangement can occur in polyhydroxyalcohols under acidic conditions, rearranging the carbon skeleton.
-
Protection of alcohols
- Trimethylsilyl (TMS) groups can protect the from unwanted reactions.
- To protect: → .
- To deprotect: add .
-
Preparation of mesylates and tosylates
- Mesylates: React with mesyl chloride ().
- Tosylates: React with tosyl chloride ().
- Both convert into a better leaving group, which makes substitution or elimination easier.
Additional transformations include reactions with to form alkyl chlorides, to form alkyl bromides, and esterification with carboxylic acids. Inorganic esters form when an alcohol reacts with non-carbon acid derivatives, such as phosphate groups in DNA/RNA polymerization, which forms phosphodiester bonds.
Carboxylic acids
Carboxylic acids contain a carboxyl group (), which drives their characteristic chemistry. They’re acidic because they can donate the proton on the .
Many naturally occurring substances are carboxylic acids, such as acetic acid in vinegar.
Nomenclature
IUPAC names typically end in “-oic acid,” though “carboxylic acid” or “-dioic acid” may be used for certain structures (e.g., ethanedioic acid for oxalic acid). Common names (like formic acid or acetic acid) are also widely used.
Physical properties and solubility
Carboxylic acids show strong hydrogen bonding, which raises their boiling points compared with compounds of similar molecular weight. They’re generally water-soluble when the alkyl chain is short; solubility decreases as the chain length increases. In IR spectroscopy, they show a broad stretch near 3100 and a sharp peak around 1700 .
Important reactions
Carboxylic acids undergo nucleophilic attack at the electrophilic carbonyl carbon. Many reactions involve replacing the (directly or indirectly) with another group, so the acid is often converted into a more reactive derivative (such as an acyl halide) before further transformations. This activation can also set up reactions like halogenation at the position.
Carboxyl group reactions
- Esterification: Under acidic conditions, a carboxylic acid reacts with an alcohol to form an ester.
- Nucleophilic attack: The carbon of the is electrophilic, so nucleophiles can add to it. The acidic proton helps by enabling proton transfers during the mechanism.
-
Amide formation: Reaction with ammonia or amines can yield amides, especially when an activated acid derivative (e.g., an acyl chloride) is used.
-
Anhydride formation: Two molecules of a carboxylic acid can link (losing water) to form an anhydride, which is more reactive toward nucleophiles.
-
Reduction
- Strong reagents like reduce a carboxylic acid to a primary alcohol. Milder reagents (e.g., ) are generally insufficient to reduce carboxylic acids.
-
Decarboxylation
- -Keto acids readily lose upon heating, breaking the bond between the carbonyl group and the carboxylate. This is facilitated by an internal cyclic transition state.
Reactions at 2 position, substitution
- The carbon (2 position) can be halogenated when an acid derivative (acyl halide) temporarily enolizes. An electrophile then adds at this enolized position, and subsequent hydrolysis reforms the carboxylic acid with the -substituent now in place.