Protein structure, non-enz protein function, lipids
Three-dimensional protein structure
Conformational stability
- Definition: A protein’s ability to maintain its functional 3D shape.
- Stabilizing forces: Hydrogen bonds, ionic attractions, hydrophobic effects, and van der Waals forces.
- Denaturation: Loss of structure under stress (e.g., heat or extreme pH), which causes the protein to unfold and lose biological activity.
- Solvation layer: Water molecules organize around hydrophobic regions. This favors a folded conformation because clustering nonpolar residues inside the protein increases net entropy.
Quaternary structure
- Association of subunits: Many proteins become functional by assembling multiple polypeptide chains. These subunits interact through the same forces that stabilize tertiary structure. Disulfide bonds may also link separate chains.
Non-enzymatic protein function
Non-enzymatic proteins carry out essential tasks without catalyzing chemical reactions. Instead, they rely on binding, immune defense, and motor functions to support key physiological roles.
Binding
Many proteins act as specialized transport or receptor molecules:
- Transport proteins: Hemoglobin binds oxygen with high specificity, delivering it efficiently through the bloodstream.
- Receptors: Located on cell surfaces, receptors recognize hormones or neurotransmitters. Binding triggers signal transduction, which leads to specific cellular responses. High-affinity interactions depend on precise structural complementarity between the receptor site and its ligand.
Immune system
Certain proteins act as antibodies, recognizing and binding antigens - distinct molecular markers on pathogens or foreign substances:
- Neutralization and tagging: By binding antigens, antibodies either neutralize potential threats or mark them for destruction by other immune cells.
- Specificity: Antibodies contain variable regions that adjust their shape to match an antigen, which helps explain the diversity of immune responses.
Motor
Some proteins convert chemical energy into mechanical work, which is essential for movement and intracellular transport:
- Myosin and actin: Myosin interacts with actin filaments to produce muscle contraction and enable cellular movement.
- Kinesin and dynein: These motor proteins move along microtubules, transporting cargo such as vesicles or organelles within cells.
Lipids - description, types
Lipids are a broad class of hydrophobic or amphipathic molecules with roles in energy storage, membrane structure, and signaling. Many lipids contain long hydrocarbon chains or ring systems, which makes them insoluble (or only partially soluble) in water.
Storage
Free fatty acids: saponification
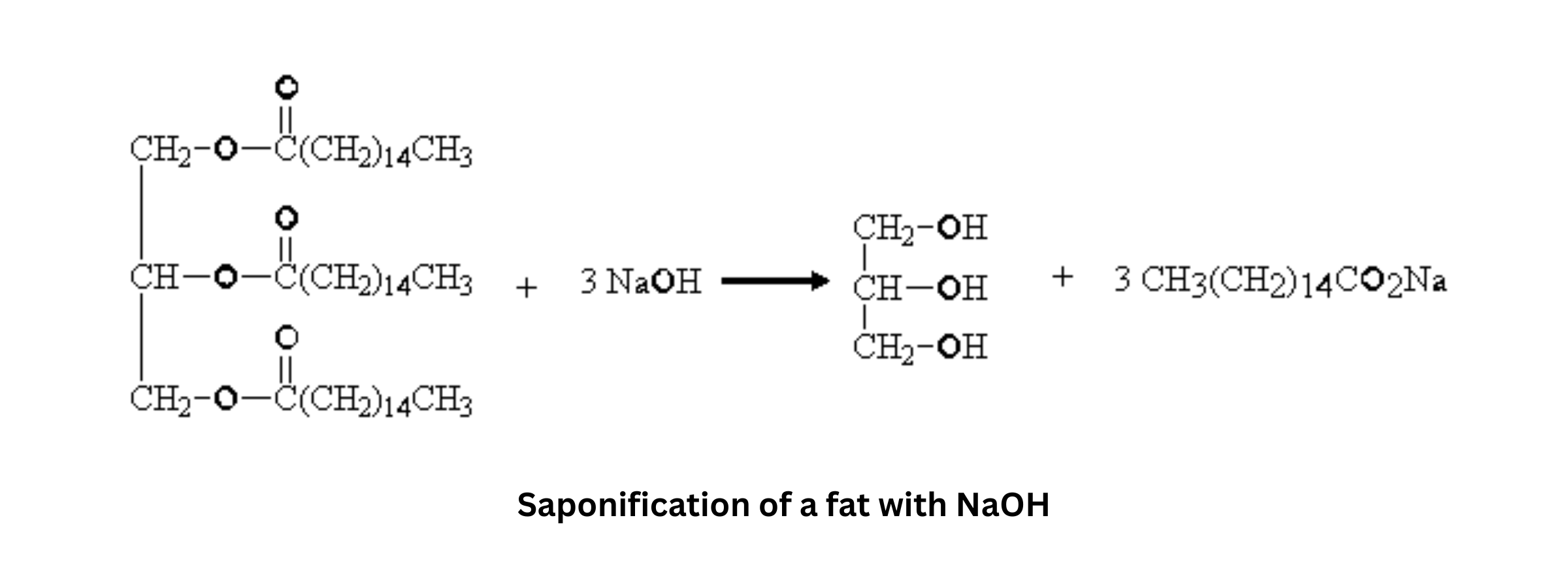
- Definition: Free fatty acids are fatty acids in their hydrolyzed form, after they’ve been cleaved from glycerol.
- Process: Saponification hydrolyzes ester bonds in fats, producing soap (the sodium or potassium salts of the fatty acids) and glycerol.
Structural
Phospholipids and phosphatids
- Major components of cell membranes, composed of two fatty acids attached to a glycerol backbone with a phosphate-containing head group.
- Their amphipathic nature drives bilayer formation, creating a selective barrier around cells.
Sphingolipids
- Derived from the amino alcohol sphingosine instead of glycerol.
- Found in neuronal membranes (e.g., myelin sheaths), where they contribute to signal transduction and cell recognition.
Waxes
- Long-chain fatty acids esterified to long-chain alcohols.
- Highly hydrophobic and typically serve protective or structural functions (e.g., on plant leaves or animal fur).
Signals/cofactors
Steroids
- Structure: Steroids (e.g., cholesterol, testosterone, estrogen) share a multi-ring system derived from the cyclization of terpene precursors.
- Biosynthesis:
- Begins with squalene, a triterpene containing six isoprene units.
- The double bonds in squalene allow it to cyclize into the core steroid nucleus.
- Function:
- Cholesterol modulates membrane fluidity and serves as a precursor for steroid hormones.
- Testosterone and estrogen act as key signaling molecules in numerous physiological processes.
Prostaglandins
- Derived from arachidonic acid through the cyclooxygenase pathway.
- Function as local signaling molecules (e.g., regulating inflammation and smooth muscle contraction).
Additional notes on terpenes
- Terpenes: Built from isoprene () subunits. Their classification depends on the number of isoprene units:
- Monoterpenes: 2 isoprene units
- Diterpenes: 4 isoprene units
- Triterpenes: 6 isoprene units (e.g., squalene)
- Cyclization: Terpenes’ double bonds enable ring formation (e.g., steroid biosynthesis from squalene).