Ions in solution, solubility, titration
Ions in solutions
Ions in solution are charged particles produced when compounds dissolve in water. There are two types:
- Anions: negatively charged ions
- Cations: positively charged ions
Refer to the textbook to view this data.
Once ions are in water, water molecules surround them in a process called hydration (also called solvation).
- The partially negative oxygen end of water is attracted to and surrounds cations.
- The partially positive hydrogen ends of water are attracted to and surround anions.
This is why a “free” H⁺ ion isn’t found in aqueous solution. Instead, it associates with water to form the hydronium ion (H₃O⁺).
Solubility
Solubility describes how much of a substance dissolves in a solvent. We often quantify solubility using concentration units:
- Molarity (M): moles of solute per liter of solution
- Molality (m): moles of solute per kilogram of solvent
- Normality (N): the effective molarity of the reactive species (e.g., )
- Percent by mass: x% = x g per 100 g or 100 mL
- Parts per million (ppm): x mg per kg or per liter
The solubility product constant () is the equilibrium constant for the dissolution of a sparingly soluble salt.
For example, the dissolution of silver chloride is:
Its equilibrium expression is:
For a salt that produces more than one ion of a given type, the exponents in the expression match the stoichiometric coefficients. For silver sulfate:
the expression is:
A higher value indicates that more of the salt dissolves.
Common-ion effect applies Le Chatelier’s principle to solubility equilibria. For example, consider:
If you add a salt containing the common ion (such as sodium chloride, ), the concentration of increases. The equilibrium shifts to the left, so less dissolves (its solubility decreases). In laboratory separations, this effect helps you selectively precipitate one component from a mixture by adding a source of an ion common to the desired precipitate.
Complex ion formation
Complex ion formation can have the opposite effect and increase solubility. When a metal ion (M⁺) binds a Lewis base (L), it forms a complex ion:
The formation constant, , quantifies how strongly the complex forms. When complex formation removes free metal ions from solution, the dissolution equilibrium for a sparingly soluble salt shifts to produce more ions, so more solid dissolves.
Solubility can also depend on pH.
For a weak acid () that dissociates as:
adding a base removes from solution. That drives the equilibrium to the right, which increases the solubility of .
Conversely, a weak base (B) dissolves more in acidic solution because added converts B to its conjugate acid:
The common-ion effect, complex ion formation, and pH adjustments are widely used in laboratory separations to selectively precipitate or dissolve compounds.
Titration
Titration is an analytical technique for finding the concentration of an unknown acid or base by reacting it with a solution of known concentration (the titrant).
Many titrations use an indicator, a compound that behaves like a weak acid or base. It’s added in such small amounts that it doesn’t significantly change the overall pH. A typical indicator equilibrium is:
with acid dissociation constant:
Because the ratio of to depends on :
- At low pH (high [H⁺]), the indicator is mostly H-In and shows one color.
- At high pH (low [H⁺]), the indicator is mostly In⁻ and shows a different color.
This color change marks the endpoint of the titration, which typically involves a neutralization reaction:
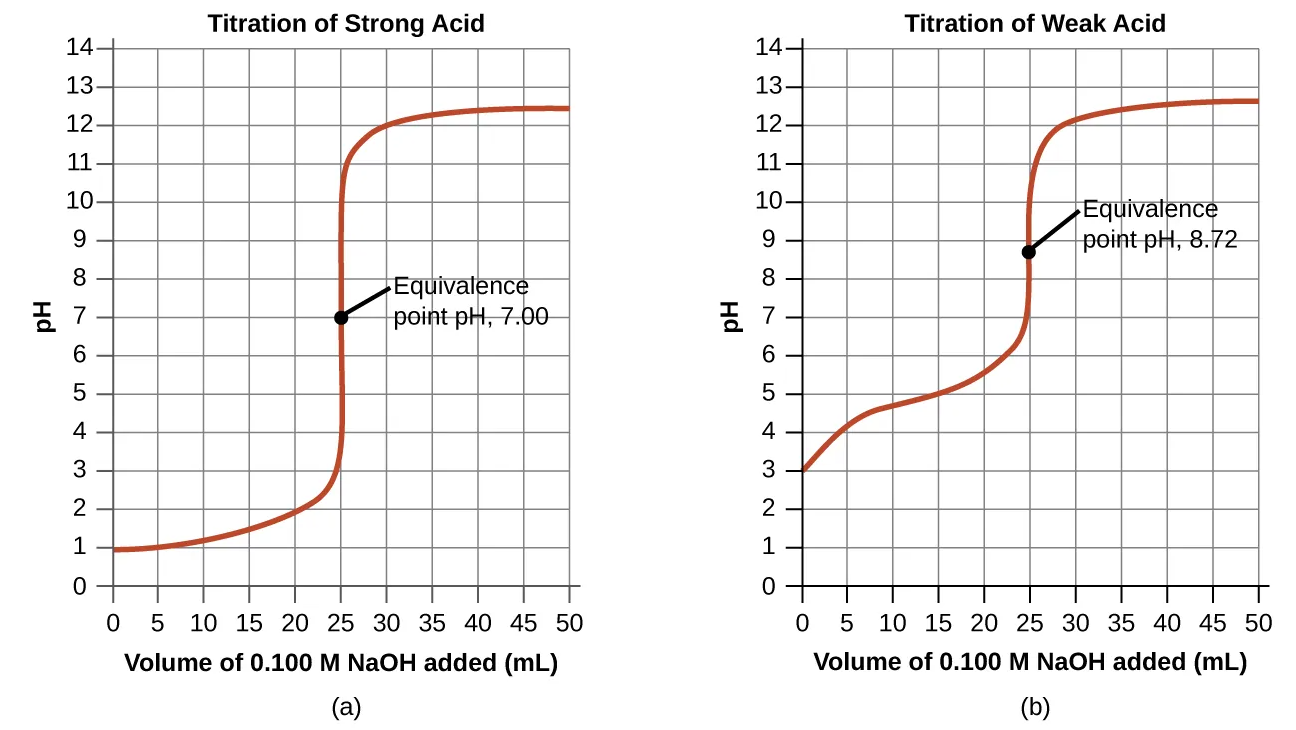
How you interpret a titration curve depends on the strengths of the acid and base. For a strong acid-strong base titration (or the reverse), the curve has a sharp transition at the equivalence point. For a weak acid-strong base titration (or a weak base-strong acid titration), a buffer region appears.
In the buffer region, the weak acid and its conjugate base (or the weak base and its conjugate acid) are present in comparable amounts. In this region, the pH is approximately equal to the pKa (or 14 minus the pKb). This buffering region usually spans about pKa ± 1.
For weak polyprotic acids (acids that can donate more than one proton), the titration curve shows multiple inflection points and equivalence points, each tied to a different pKa. At each inflection point, the concentration of an acidic species equals the concentration of its conjugate base, which allows you to determine the individual dissociation constants.
In summary, titration tracks pH changes during neutralization. Indicators and buffer behavior help you identify key points on the curve and determine the concentration of the analyte.