Acid/base equilibria
Acid/base equilibrium is a reversible reaction between a Brønsted acid (a proton donor) and a Brønsted base (a proton acceptor). When the acid donates a proton, it becomes its conjugate base. When the base accepts a proton, it becomes its conjugate acid.
Ionization of water
A classic example is the ionization of water:
The product of the hydrogen ion concentration () and the hydroxide ion concentration () is the ion-product constant for water, , which is approximately at C.
The pH of a solution is defined as the negative logarithm of the hydrogen ion concentration:
For pure water, , so the pH is , which indicates a neutral solution.
A solution is acidic if its pH is less than and basic if its pH is greater than . The pOH is defined as , and at C:
.
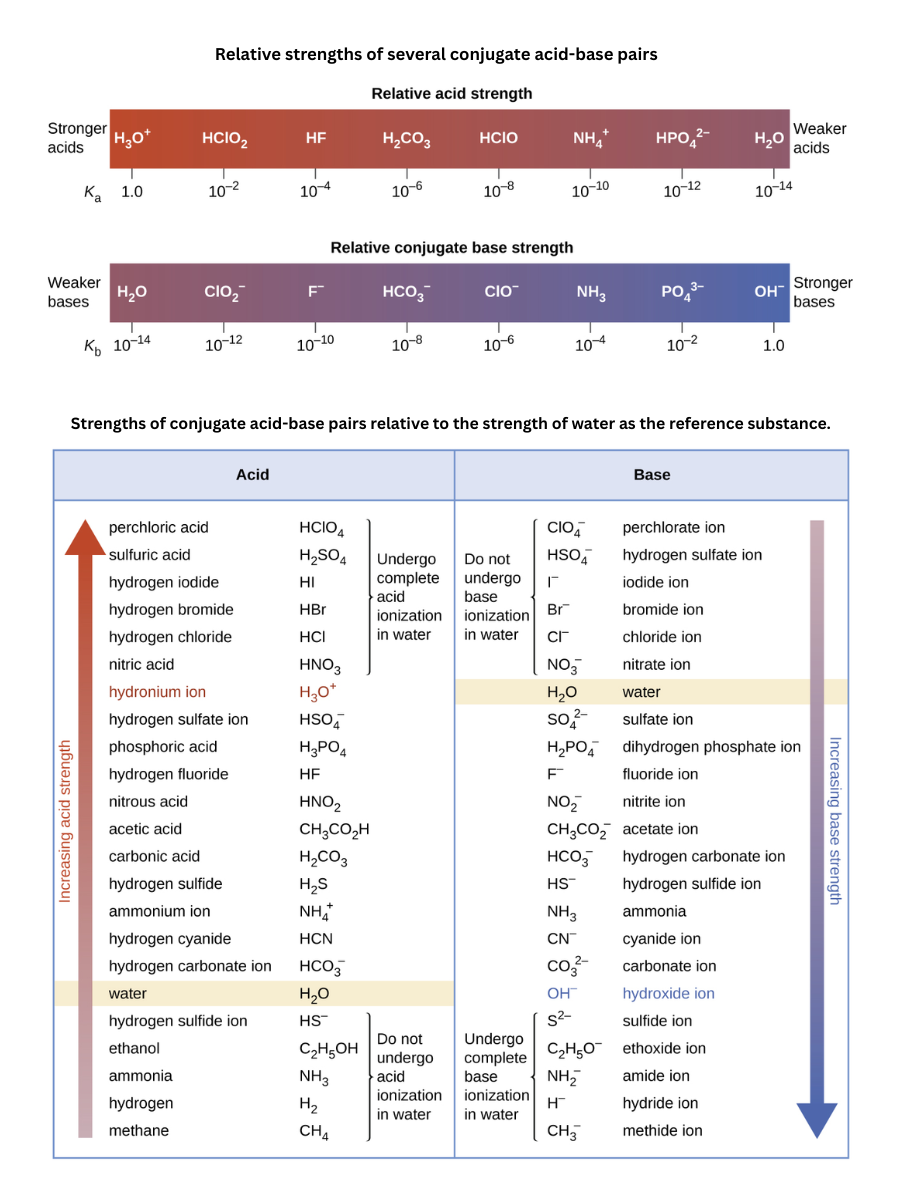
Conjugate acids and bases (e.g., and )
Acid/base equilibria describe how acids and bases interconvert with their conjugate acids and conjugate bases. The key idea is simple:
- When an acid donates a proton, it forms its conjugate base.
- When a base accepts a proton, it forms its conjugate acid.
- In strong acids and strong bases, complete dissociation occurs because their conjugate species are highly stable.
- Weak acids and weak bases only partially dissociate due to the moderate stability of their conjugate species.
- This equilibrium can be shifted by the presence of a common ion, as explained by Le Chatelier’s principle; for instance, acetic acid () dissociates less in the presence of its salt, , and ammonium hydroxide () shows reduced dissociation when is added.
Calculation of pH of solutions of salts of weak acids or bases
Consider a 0.10 M aqueous solution of sodium acetate (). In water, sodium acetate dissociates completely into and (the acetate ion). The relevant equilibrium is the hydrolysis of the acetate ion:
Acid/base constants:
Acetic acid () has a dissociation constant of . Because is the conjugate base of a weak acid, its basicity is described by , which we find using:
Setting up the equilibrium expression:
Let be the concentration of produced by acetate hydrolysis. The equilibrium expression for the reaction is:
Initially,
At equilibrium,
Because is very small, we approximate:
Substituting into the equilibrium expression gives:
Solving for :
Therefore,
Calculating pOH and pH:
Hence, a solution of sodium acetate is basic, with a . This happens because the acetate anion acts as a weak base and partially hydrolyzes in water, producing and raising the pH above .
Equilibrium constants and : ,
The acid dissociation constant () describes how much an acid donates protons () in water. A general form is:
Similarly, the base dissociation constant () describes how strongly a base produces hydroxide by accepting a proton from water:
Note that water is not included in these expressions because its concentration remains constant.
For a conjugate acid-base pair, the product of and equals the ionization constant of water (), typically . To make these constants easier to compare, we define pKa and pKb as the negative logarithms of and , respectively:
and for any conjugate pair,
Buffers
Buffers are solutions that resist changes in pH by maintaining an equilibrium between an acidic species and its conjugate base. In a buffer:
- the acidic component donates protons when the pH rises
- the basic component accepts protons when the pH falls
Buffers are typically composed of salts derived from weak acids and weak bases. Maximum buffering capacity occurs when the concentrations of the acid and its conjugate base are equal. In that case, the pH equals the of the acid (or, for a weak base, the pH equals minus the ).
Influence on titration curves
On a titration curve, the buffering region appears as a relatively flat segment near the point of inflection. It typically spans about one pH unit above and below the (or ).