The periodic table
The periodic table - classification of elements into groups by electronic structure
Alkali metals
The periodic table groups elements by electronic structure, which helps explain why elements in the same group share similar physical and chemical properties. For example, alkali metals have a single valence electron. Because that outer electron is relatively easy to remove, alkali metals have low ionization energy and are highly reactive.
Alkali metals typically lose their valence electron to reach a more stable electron configuration, so they most often form compounds in the +1 oxidation state. Their reactivity increases down the group as atomic radii increase and the valence electron is held less tightly. They commonly react:
- With oxygen to form oxides
- With water to form hydroxides and release hydrogen gas
- With acids to form salts and release hydrogen gas
Alkaline earth metals
Alkaline earth metals have two valence electrons. They also have relatively low ionization energies, but they’re generally less reactive than alkali metals because removing two electrons requires more energy than removing one.
These elements tend to lose both valence electrons to reach a stable electron configuration, so they commonly form compounds in the oxidation state. Their reactivity increases as atomic radii increase down the group, and they react with oxygen, water, and acids in predictable patterns.
Halogens and noble gases
The halogens have 7 valence electrons (2 in the s subshell and 5 in the p subshell). This makes them strongly attracted to gaining one more electron, so they have high electron affinity and are very reactive. Halogens typically gain one electron to complete the valence shell, so they commonly appear in the -1 oxidation state.
Halogen reactivity increases as you move up the group because atomic radii decrease and the nucleus attracts an added electron more strongly. Halogens readily react with alkali and alkaline earth metals to form salts.
In contrast, the noble gases have a full valence shell of 8 electrons. This stable configuration corresponds to high ionization energy and low electron affinity, so noble gases are virtually non-reactive and are generally found in the 0 oxidation state.
Transition metals and representative elements
Transition metals are good conductors because they have loosely bound d electrons that can move through the metal.
In chemical complexes, ligands bind to transition metals and cause the (formerly degenerate) d orbitals to split into nondegenerate energy levels. Electrons can transition between these split d orbitals, which produces the vivid colors seen in many transition metal compounds. Transition metals also show a range of (typically positive) oxidation states, which contributes to their diverse chemistry.
In contrast, representative elements (the s and p blocks) do not have free-moving d electrons. Their valence shells fill in a more predictable way, from 1 to 8 valence electrons across the main-group columns (IA, IIA, IIIA, etc.).
Metals and non-metals
On the periodic table, metals lie to the left of the metalloids, while non-metals lie to the right.
Chemically, metals tend to lose electrons and form positive oxidation states, which makes them effective reducing agents. Metals have low electronegativity, so in covalent bonds with non-metals they often carry partial positive character. They also tend to form basic oxides.
Physically, metals:
- Conduct heat and electricity well
- Are malleable and ductile
- Have a characteristic luster
- Are typically solid at room temperature (except mercury)
Non-metals tend to gain electrons, often forming negative oxidation states. Because they have higher electronegativity, they are strong oxidizing agents and tend to form acidic oxides.
Physically, non-metals may be solid, liquid, or gas at room temperature. When solid, they are usually brittle and lack metallic luster.
Oxygen group
The oxygen group (the chalcogens) is the column that begins with oxygen. Oxygen and sulfur are both non-metals and show similar chemical reactivity, which is why sulfur can substitute for oxygen in some reactions.
Moving down the group, properties shift gradually:
- Selenium remains a non-metal
- Tellurium behaves as a metalloid
- Polonium shows metallic characteristics or is considered a metalloid
This pattern matches the broader periodic trend: metals are generally found to the left of metalloids, and changes in atomic structure across the table influence both reactivity and physical properties.
The periodic table - variations of chemical properties with group and row
An atom’s electronic structure changes in systematic ways across the periodic table, and those changes drive trends in chemical properties.
Representative elements (the s block and p block) do not have free-flowing outer d electrons. Their valence shell fills from one electron on the far left to eight electrons on the far right, and these elements are labeled using standard group nomenclature (IA, IIA, IIIA, etc.). For example, the noble gases have a complete valence shell of eight electrons, which corresponds to high ionization energy and low electron affinity. As a result, they are chemically inert and typically found in the 0 oxidation state.
Transition metals, on the other hand, have loosely bound outer d electrons. This gives them high conductivity and flexible bonding behavior. In complexes, ligands cause the metal’s d orbitals to split into nondegenerate energy levels. Electron transitions between these levels produce the vivid colors seen in many transition metal compounds. Transition metals also commonly show multiple positive oxidation states because their d electrons can participate in bonding in different ways.
Valence electrons
Valence electrons are the electrons in an atom’s outermost shell that most strongly influence chemical behavior.
In representative elements, the number of valence electrons increases from one to eight as you move left to right across a period. This simple pattern does not apply to transition metals, where electrons in inner d orbitals also affect chemical properties.
First and second ionization energy
The first ionization energy is the energy required to remove the first valence electron from an atom. The second ionization energy is the energy required to remove a second electron after the first has been removed. Both depend on the atom’s electronic structure.
Noble gases have the highest ionization energies because their valence shells are full. Alkali metals have the lowest because their single valence electron is relatively easy to remove. You’ll also see local maxima in ionization energy for elements with filled or half-filled subshells, since those arrangements are especially stable.
The second ionization energy is always much higher than the first. This difference is especially dramatic for:
- Alkali metals, where removing the first electron is easy but removing the next electron disrupts a much more stable configuration
- Hydrogen, where there is no second electron to remove after the first is removed
In alkaline earth metals, both the first and second ionization energies are relatively low, consistent with their tendency to lose two valence electrons.
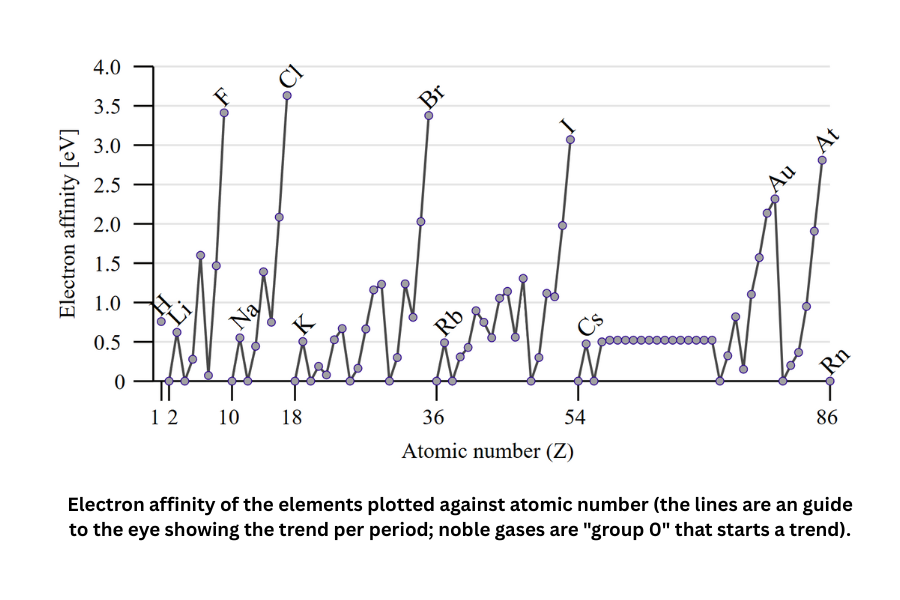
Electron affinity
Electron affinity is the energy released when an atom gains an electron. It reflects how strongly an atom tends to accept an electron.
Electron affinity generally decreases down a group because increasing atomic radii weaken the attraction between the nucleus and the added electron. Across a period from left to right, electron affinity tends to increase because atomic radius decreases and the effective nuclear charge pulls more strongly on the incoming electron.
The halogens have the highest electron affinities, while the noble gases have the lowest. Local minima occur for atoms with filled subshells or half-filled p subshells, which are especially stable and therefore less likely to release energy when gaining an electron.
Electronegativity
Electronegativity measures an atom’s ability to attract electrons within a covalent bond. An atom with higher electronegativity pulls electron density toward itself and takes on a partial negative charge. An atom with lower electronegativity (more electropositive) pulls less electron density and takes on a partial positive charge.
Electronegativity generally increases toward the top right of the periodic table. Fluorine is the most electronegative element. Elements near fluorine - such as nitrogen, oxygen, chlorine, and bromine - are also highly electronegative, which is typical of halogens.
Non-metals are usually more electronegative than metals. Some noble gases, such as krypton and xenon, can show significant electronegativity when they form bonds.
When the electronegativity difference between two atoms becomes large enough, the bond is no longer covalent. Instead, it becomes ionic, formed by the transfer of electrons from the electropositive atom to the electronegative atom.
Electron shells and the sizes of atoms
Electron shells are regions around the nucleus defined by the principal quantum number ().
- Moving down the periodic table adds a new electron shell, which increases the atomic radius. For example, when moving from neon to sodium, the outer electrons feel a reduced effective nuclear charge because inner electrons shield them from the full positive charge of the nucleus.
- Moving down a group (for example, sodium to potassium) produces larger atoms because electrons are added to shells farther from the nucleus, even though the effective nuclear charge stays relatively constant.
- Moving across a period adds electrons to the same shell. Because these electrons do not shield each other effectively, the effective nuclear charge increases. The stronger nuclear pull increases the electrostatic attraction (F = kQq/r²) between the nucleus and the electrons, pulling electrons closer and decreasing atomic size.
Electron shells and the sizes of ions
The size of an ion depends on its number of electrons and the effective nuclear charge.
When an atom loses electrons to form a cation, electron-electron repulsion decreases while nuclear charge stays the same, so the ion is smaller than the neutral atom. When an atom gains electrons to form an anion, electron-electron repulsion increases, so the ion is larger than its parent atom.