Stoichiometry
Molecular weight and formula, common metric units in chemistry
Molecular weight is numerically equivalent to the molecular mass of a substance and is measured in amu. Since 1 amu is defined as 1 , an element like Oxygen-16, with a mass of 16 amu, has a molar mass of 16 .
The molecular formula of a compound (for example, glucose, ) gives the exact number of each type of atom in one molecule. The empirical formula gives the simplest whole-number ratio of those atoms (for glucose, ).
In chemistry, you’ll often see these metric units:
- Molarity (): moles of solute per liter of solution (a concentration unit)
- Molality (): moles of solute per kilogram of solvent (also a concentration unit)
- Molar mass: typically expressed in
- Mass: commonly measured in kilograms (kg)
Composition by percent mass, mole concept, density
- % Mass represents the proportion of a specific species’ mass relative to the total mass of a sample, calculated as (% mass = [mass of species / total mass] × 100).
- The mole concept lets you count particles (atoms, molecules, ions) using a macroscopic amount of material: 1 mole equals Avogadro’s number (approximately molecules).
- Density is defined as mass per unit volume (density = mass/volume, measured in ). In chemistry, specific gravity is often used instead. Specific gravity is the ratio of the density of a substance to that of water (density of water = 1 or 1 ), so it has no units. For example, if lead has a density of 11 , its specific gravity is 11.
Oxidation number
The oxidation number of an atom is the charge it would carry if the compound were entirely composed of ions.
Oxidizing agents are substances that gain electrons during a redox reaction, causing another substance to be oxidized. Common examples include oxygen (), ozone (), permanganates (), chromates (), dichromates (), and peroxides (), as well as various Lewis acids or oxygen-rich compounds. These agents have a high electron affinity and typically increase their oxidation state by accepting electrons.
In contrast, reducing agents donate electrons, so they reduce other species. Typical reducing agents include hydrogen (), reactive metals such as potassium, and reducing mixtures like and , along with strong reductants like LAH (Lithium Aluminum Hydride) and (Sodium Borohydride), as well as various Lewis bases or hydrogen-rich compounds. These agents tend to lower their oxidation state by losing electrons.
An important redox pattern is disproportionation, where the same element in a single oxidation state is both oxidized and reduced. For example, in the reaction , the ion plays both roles:
- One loses an electron to become (oxidation).
- The other gains an electron to form elemental copper () (reduction).
Redox titration
Redox titration is an analytical technique used to determine the concentration of an unknown analyte by reacting it with a known titrant in a redox (reduction-oxidation) reaction. In these titrations:
- The analyte may be in a reduced form () or an oxidized form ().
- The titrant is chosen as either a strong oxidizing agent () or a reducing agent (), depending on what will react with the analyte.
- A standard solution (with accurately known concentration) is often used so the amount of titrant added can be related precisely to the amount of analyte present.
A common variant is iodimetric titration, where the redox chemistry involves iodine (). For example, a reduced analyte can react with iodine to form its oxidized form and iodide ions (). In more complex procedures, an intermediate species () may form; it can exist as or and later reacts with the titrant.
The endpoint is typically detected by a clear color change. For instance, iodine forms a dark blue complex with starch; when all iodine has been reduced, the blue color disappears, indicating the reaction is complete. This is analogous to acid-base titration, but the key change being tracked is oxidation state rather than pH.
Description of reactions by chemical equations
A chemical equation follows conventions that show the reactants and products and their phases - (s) for solids, (l) for liquids, (g) for gases, and (aq) for aqueous solutions.
- Coefficients are placed before each compound to balance the equation, meaning the number of atoms of each element is the same on both sides.
- The arrow shows the direction of the reaction: a single-headed arrow indicates a reaction that goes to completion, while a double-headed arrow indicates equilibrium. If one side of the double arrow is larger, the equilibrium favors that side.
- The charge on ions is shown with symbols such as + or -, while neutral species are written without a charge.
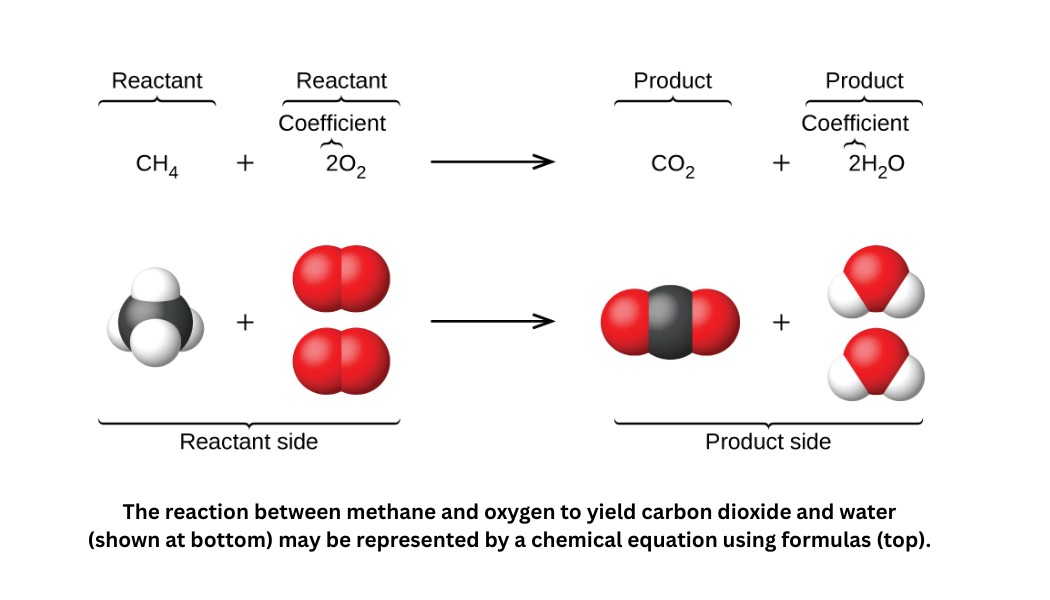
A balanced chemical equation for the combustion of methane
To balance chemical reactions:
- start by balancing each of the half reactions separately, ensuring that both the atoms and charge are equal on both sides.
- Under acidic conditions, balance oxygen atoms by adding to the side that needs oxygen, and then add to the opposite side to balance the added hydrogen atoms.
- Under basic conditions, add to the side lacking oxygen and then add to the other side to balance the hydrogens. Two common methods used are the Ion-Electron Method, which balances atoms first and then charge, and the Oxidation-State Method, where you treat the species undergoing a change in oxidation number as a single entity.
- Once the half reactions are balanced, multiply them by appropriate factors so that the electrons cancel out when you add the equations together.
- Finally, combine identical species on the same side, cancel out any species that appear on both sides, and add back any spectator ions. Ensure that the final equation has the same number of each atom on both sides and a neutral net charge.
Limiting reactants and theoretical yields
The limiting reactant is the substance that is completely consumed first in a chemical reaction. Because it runs out first, it sets the maximum amount of product that can form.
The theoretical yield is the maximum amount of product predicted by stoichiometry under ideal conditions. To find it, identify the limiting reactant and then use the balanced equation to convert from the amount of limiting reactant to the expected amount of product.
The percent yield compares what you actually obtained to what stoichiometry predicts:
- Experimental yield: the amount of product actually collected
- Percent yield: (experimental yield ÷ theoretical yield) × 100
In practice, the experimental yield is usually lower than the theoretical yield because some product is lost or side reactions occur.
Calculation of percent yield
Upon reaction of 1.274 g of copper sulfate with excess zinc metal, 0.392 g copper metal was obtained according to the equation:
What is the percent yield?
The provided information identifies copper sulfate as the limiting reactant, so we use it to calculate the theoretical yield (as illustrated in the previous module):
Using this theoretical yield and the given actual yield, calculate percent yield:
Adapted from Example 7.13, OpenStax