Citric acid cycle, ETP, OP, hormonal regulation
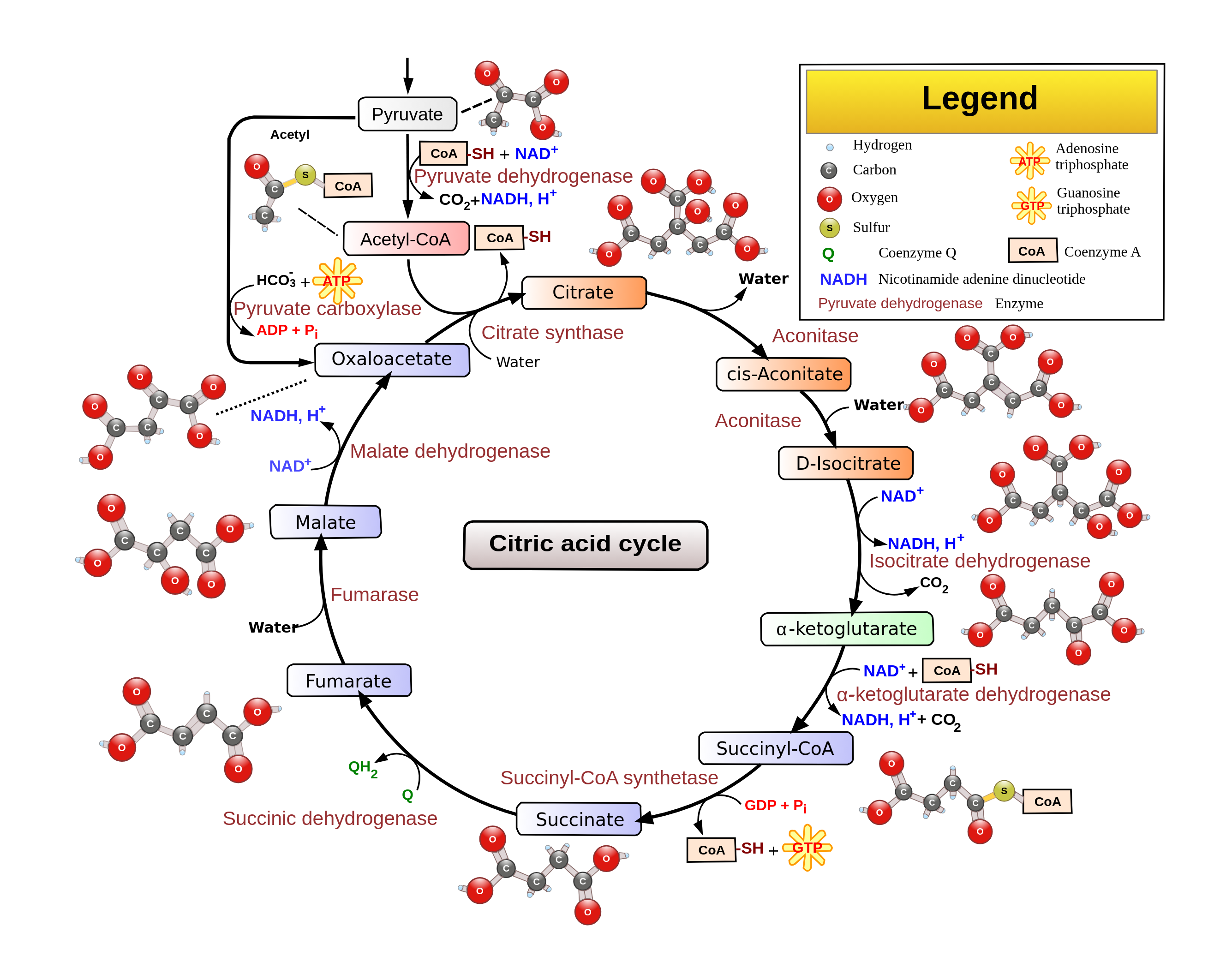
The Krebs cycle (TCA, citric acid cycle)
The Krebs cycle, also called the TCA cycle or citric acid cycle, occurs in the mitochondrial matrix. Acetyl CoA (produced from several metabolic pathways) enters the cycle and is processed through a series of reactions that fully oxidize the acetyl group to carbon dioxide.
For each acetyl CoA that enters the cycle, the cell produces:
- Three NADH
- One FADH₂
- One ATP (or GTP)
During the cycle’s early steps, coenzyme A is regenerated, which allows additional acetyl CoA molecules to enter and keep the cycle running.
The Krebs cycle is tightly regulated. When ATP and NADH levels are high (signaling that the cell’s energy needs are being met), key steps in the cycle are inhibited, helping maintain energy homeostasis.
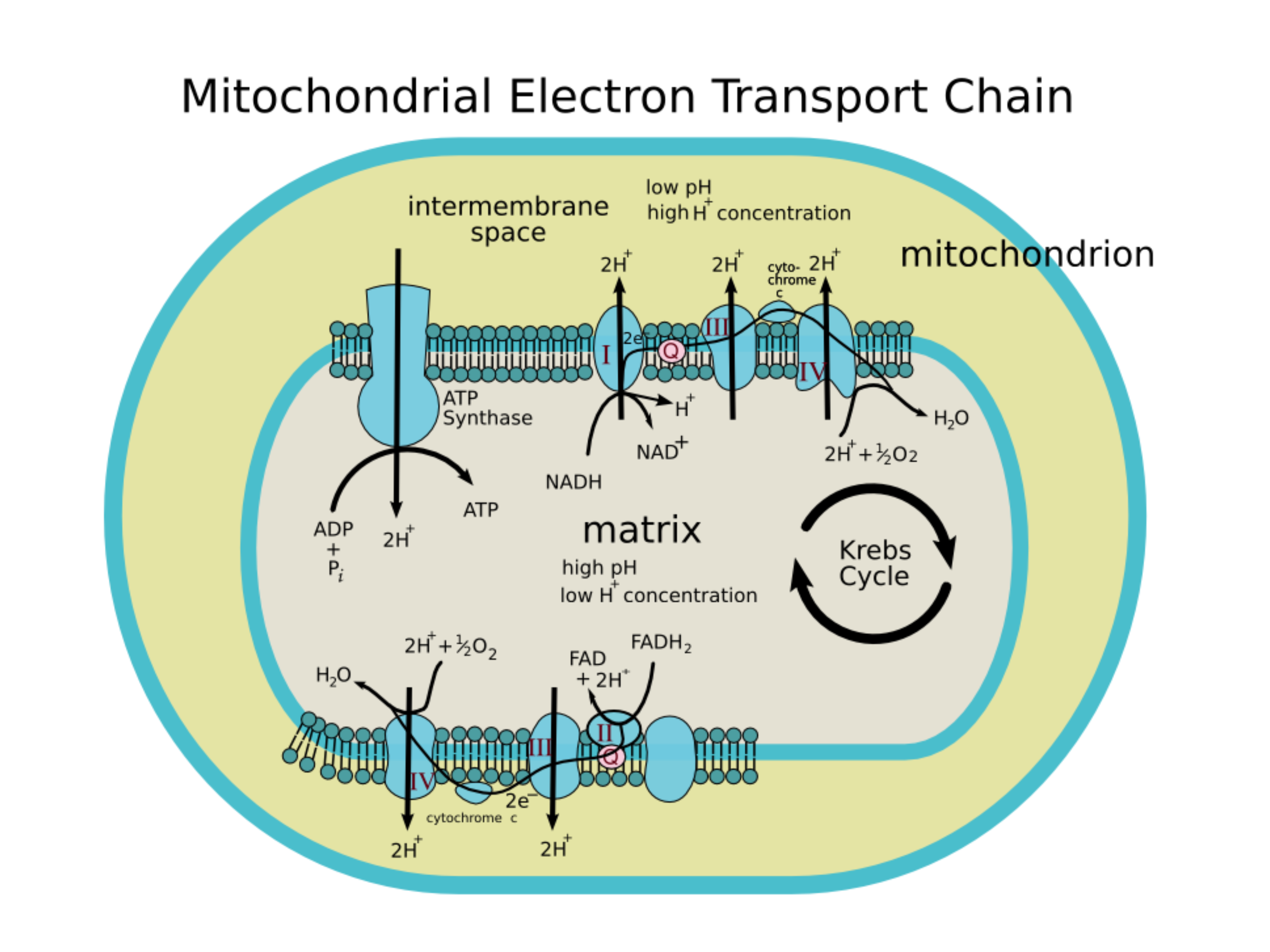
Electron transport chain (ETC) and oxidative phosphorylation
The electron transport chain (ETC) and oxidative phosphorylation occur on the inner mitochondrial membrane, along the cristae. High-energy electrons carried by and (produced largely by the Krebs cycle) move through a series of redox reactions involving FMN, Coenzyme Q, iron-sulfur clusters, and cytochromes (including cytochrome b, c, and aa₃).
As electrons flow from (relatively high energy) to oxygen (the final electron acceptor, relatively low energy), energy is released. The ETC uses that energy to pump protons (H⁺) from the mitochondrial matrix into the intermembrane space, creating an electrochemical proton gradient.
That gradient (the proton motive force) powers ATP synthase. As protons move back into the matrix through ATP synthase, the enzyme uses the released energy to convert ADP into ATP. This ATP-producing step is called oxidative phosphorylation.
The ETC can be disrupted by inhibitors, including some antibiotics, cyanide, azide, and carbon monoxide.
Although is the primary electron donor for the mitochondrial ETC, NADPH is also important. Produced mainly by the pentose phosphate pathway, NADPH supplies reducing power for biosynthetic reactions and antioxidant defenses. These roles help maintain cellular redox balance and can support electron transport efficiency under stress.
Flavoproteins are enzymes that contain flavin cofactors (FAD or FMN). These cofactors allow flavoproteins to participate in redox reactions by accepting and donating electrons. For example, in Complex II of the ETC, FAD accepts electrons during succinate oxidation and transfers them to ubiquinone, linking the citric acid cycle with oxidative phosphorylation for ATP production.
Oxidative phosphorylation
- Oxidative phosphorylation is the process by which mitochondria convert electron energy into ATP. ATP synthase generates ATP as protons flow down their concentration gradient, a mechanism called chemiosmotic coupling. The ETC pumps protons from the mitochondrial matrix into the intermembrane space, creating a proton motive force (PMF) that drives ATP synthesis as protons return to the matrix.
- Under optimal conditions, the complete oxidation of one glucose molecule yields about 30-32 ATP, reflecting the tight coupling between the ETC and ATP synthesis. This process is regulated by factors such as ADP availability, substrate levels, oxygen concentration, and ATP feedback.
- Mitochondria also influence cell fate via apoptosis. Disruptions in oxidative phosphorylation can increase reactive oxygen species (ROS) production, contributing to oxidative stress and cell death.
Metabolism of fatty acids and proteins
Metabolism of fats
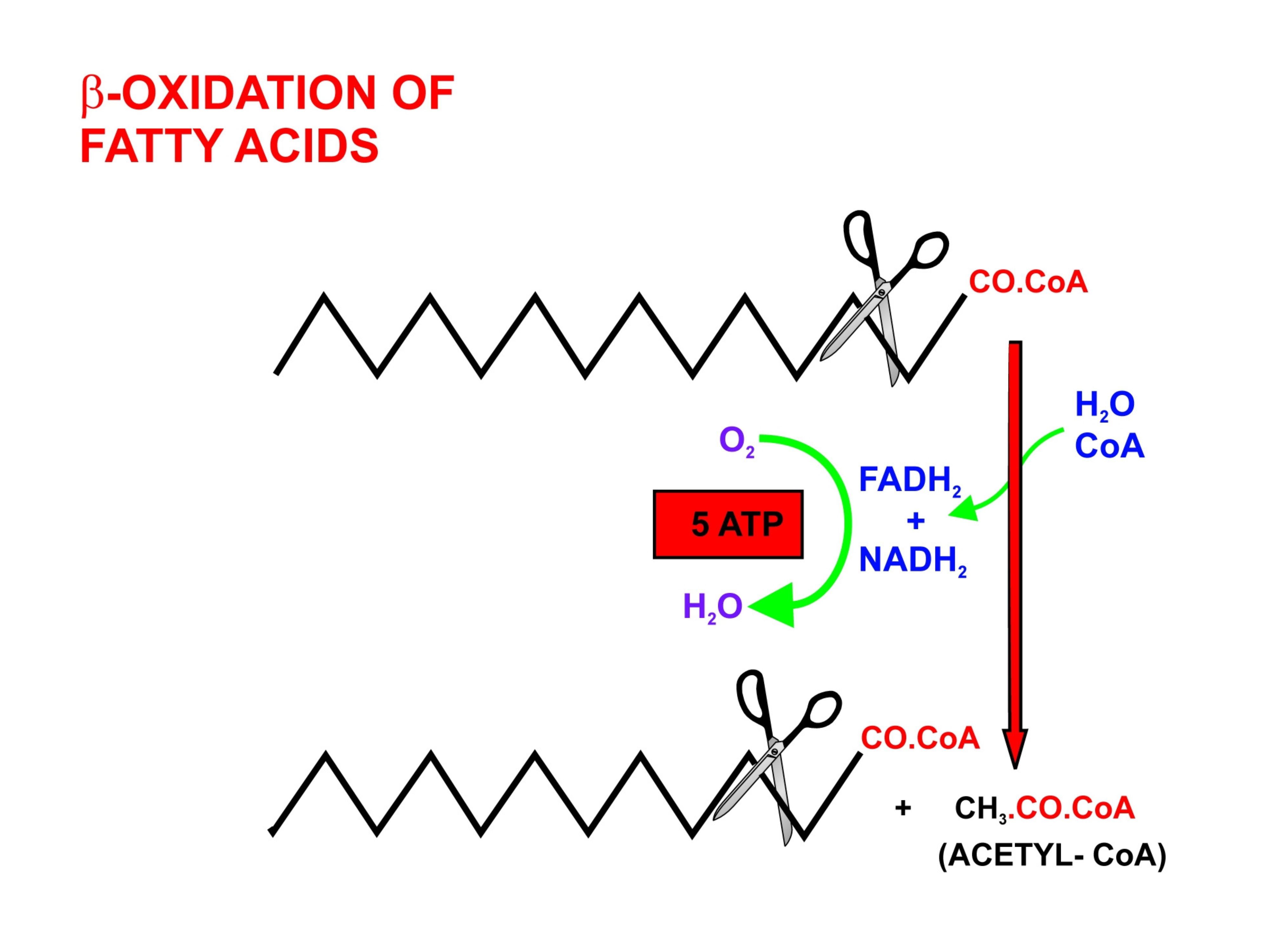
- Fats are digested in the small intestine. Their components are packaged into chylomicrons for transport through the lymphatic system and bloodstream, delivering lipids to tissues. In cells, ester hydrolysis by lipases in the cytosol converts triglycerides into free fatty acids and glycerol. Each fatty acid is then “activated” by forming a thioester bond with CoA, a reaction that requires ATP.
- In -oxidation, these acyl-CoA molecules are sequentially cleaved into two-carbon units of acetyl-CoA, generating and in the process. Acetyl-CoA enters the Krebs cycle, while and deliver electrons to the ETC, ultimately driving ATP synthesis. Per gram, fats yield more energy than any other macronutrient.
- In the liver, when carbohydrate availability is low, excess acetyl-CoA can be converted into ketone bodies (acetoacetate, -hydroxybutyrate), which serve as alternative energy sources for tissues such as the brain.
Saturated and unsaturated fats
- Saturated fatty acids have no carbon-carbon double bonds
- Unsaturated fatty acids contain one or more double bonds, affecting their fluidity and metabolic processing.
Anabolism/synthesis of fats
- Cells don’t only break down fats; they can also perform anabolism of fats, synthesizing fatty acids and triglycerides from acetyl-CoA. This is a form of non-template synthesis, where complex biomolecules like lipids (and polysaccharides) are built without following a nucleic-acid-encoded template. These pathways support energy storage, membrane structure, and signaling.
Metabolism of proteins
- Proteins are hydrolyzed into amino acids by peptidases. The amino groups are removed and converted into nitrogenous waste such as urea (or uric acid in certain species).
- The remaining carbon skeleton can be converted into pyruvate, acetyl-CoA, or other metabolic intermediates, depending on the amino acid. These carbon skeletons can enter the Krebs cycle or serve as substrates for gluconeogenesis, linking protein catabolism to the cell’s energy and biosynthetic needs.
Hormonal regulation and integration of metabolism
Hormones help coordinate metabolism by signaling when to store fuel and when to mobilize it, so tissues receive energy when they need it. Both hormone structure (peptide, steroid, or amino-acid derived) and tissue-specific metabolism influence how cells respond.
For example, the pancreas secretes insulin (a peptide hormone) when blood glucose rises. Insulin promotes glucose uptake - especially in muscle and adipose - and encourages storage as glycogen or fat. In contrast, glucagon (also secreted by the pancreas) rises when blood glucose is low and stimulates glycogenolysis and gluconeogenesis in the liver, increasing circulating glucose.
Epinephrine and cortisol
- Further integration comes from hormones like
- Epinephrine (adrenaline), which accelerates glycogen breakdown in muscle during acute stress
- Cortisol, a steroid hormone that increases protein and fat catabolism and upregulates gluconeogenesis over longer periods.
Individual tissues thus maintain specialized roles:
- Muscle primarily uses glucose for rapid ATP production
- Adipose stores excess energy as triglycerides
- The liver coordinates overall fuel distribution, converting substrates (e.g., amino acids, lactate, glycerol) into glucose or ketone bodies as needed.
Hormonal regulation of fuel metabolism
- Hormonal regulation also shapes fuel metabolism across feeding, fasting, and exercise states. In the fed state, insulin predominates, promoting glucose uptake and storage. During fasting or exercise, glucagon and epinephrine rise, mobilizing glycogen and fatty acids to maintain blood glucose and energy output.
Obesity and regulation of body mass
- Chronic energy imbalance can lead to obesity, characterized by increased adipose tissue mass. This can contribute to hormonal dysregulation, including reduced insulin sensitivity and metabolic syndrome.
- Both genetic factors and hormonal feedback loops (e.g., involving leptin and ghrelin, which regulate hunger and satiety) contribute to regulating body mass, highlighting how integrated hormonal signals can maintain - or disrupt - energy homeostasis.