Glycolysis, gluconeogenesis, metabolic regulation
Glycolysis, gluconeogenesis, and the pentose phosphate pathway
Metabolism includes all the chemical processes that keep an organism alive. It has two complementary sides:
- Catabolism: breaking down molecules to release energy
- Anabolism: using energy to build and store complex molecules
In aerobic metabolism, oxygen is required to completely oxidize a fuel source - commonly glucose - into carbon dioxide and water, while capturing released energy as ATP. Under these conditions, one molecule of glucose typically yields around 30 ATP. The overall reaction is often summarized as:
- Glucose (), obtained from the diet, provides carbons and hydrogens.
- (molecular oxygen) serves as the final electron acceptor in the electron transport chain, ultimately forming water.
- is released as the carbons (and original oxygens) from glucose are removed in processes like the Krebs cycle.
- forms in the final stages of the electron transport chain, when electrons and protons combine with inhaled .
Glycolysis
Glycolysis is a central metabolic pathway that breaks glucose down into pyruvate, producing small amounts of ATP and NADH.
Under aerobic conditions, pyruvate enters the mitochondria, where it supports further energy production through the citric acid cycle and oxidative phosphorylation.
Steps in glycolysis:
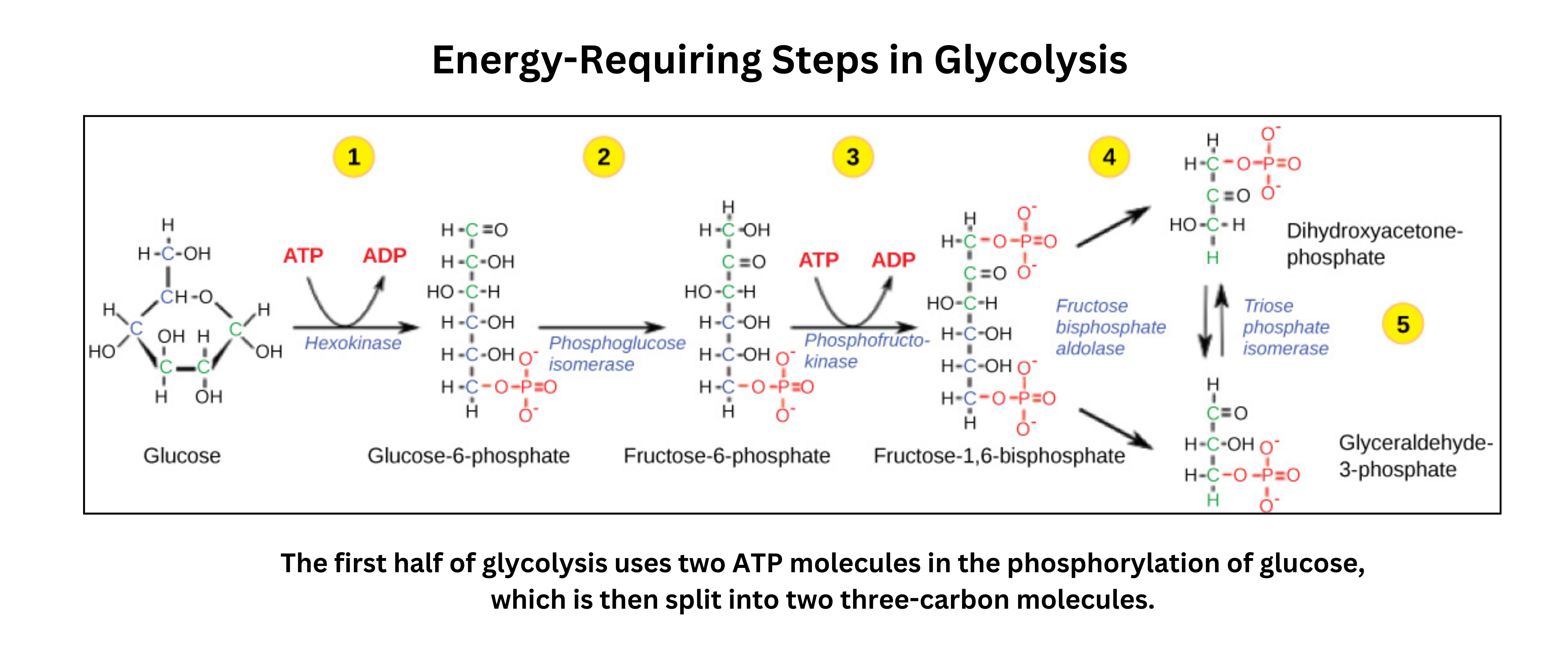
- Hexokinase phosphorylation
Glucose () enters the cell and is phosphorylated by hexokinase. This uses one ATP to form glucose-6-phosphate. Phosphorylation traps glucose inside the cell and makes it more reactive. - Isomerization
An isomerase converts glucose-6-phosphate into fructose-6-phosphate. This rearrangement prepares the molecule for another phosphorylation and for later splitting into two three‑carbon units. - Second Phosphorylation
Phosphofructokinase phosphorylates fructose-6-phosphate, using a second ATP, to produce fructose‑1,6-bisphosphate. This enzyme is a key control point in glycolysis and responds to cellular ATP/ADP levels. - Sugar Cleavage
Aldolase splits fructose-1,6-bisphosphate into two three-carbon molecules: dihydroxyacetone phosphate (DHAP) and glyceraldehyde‑3‑phosphate (G3P). - Isomerization to G3P
An isomerase converts DHAP into glyceraldehyde‑3‑phosphate, so the pathway continues with two G3P molecules. By this point, two ATP have been invested per glucose. - Oxidation and Phosphate Addition
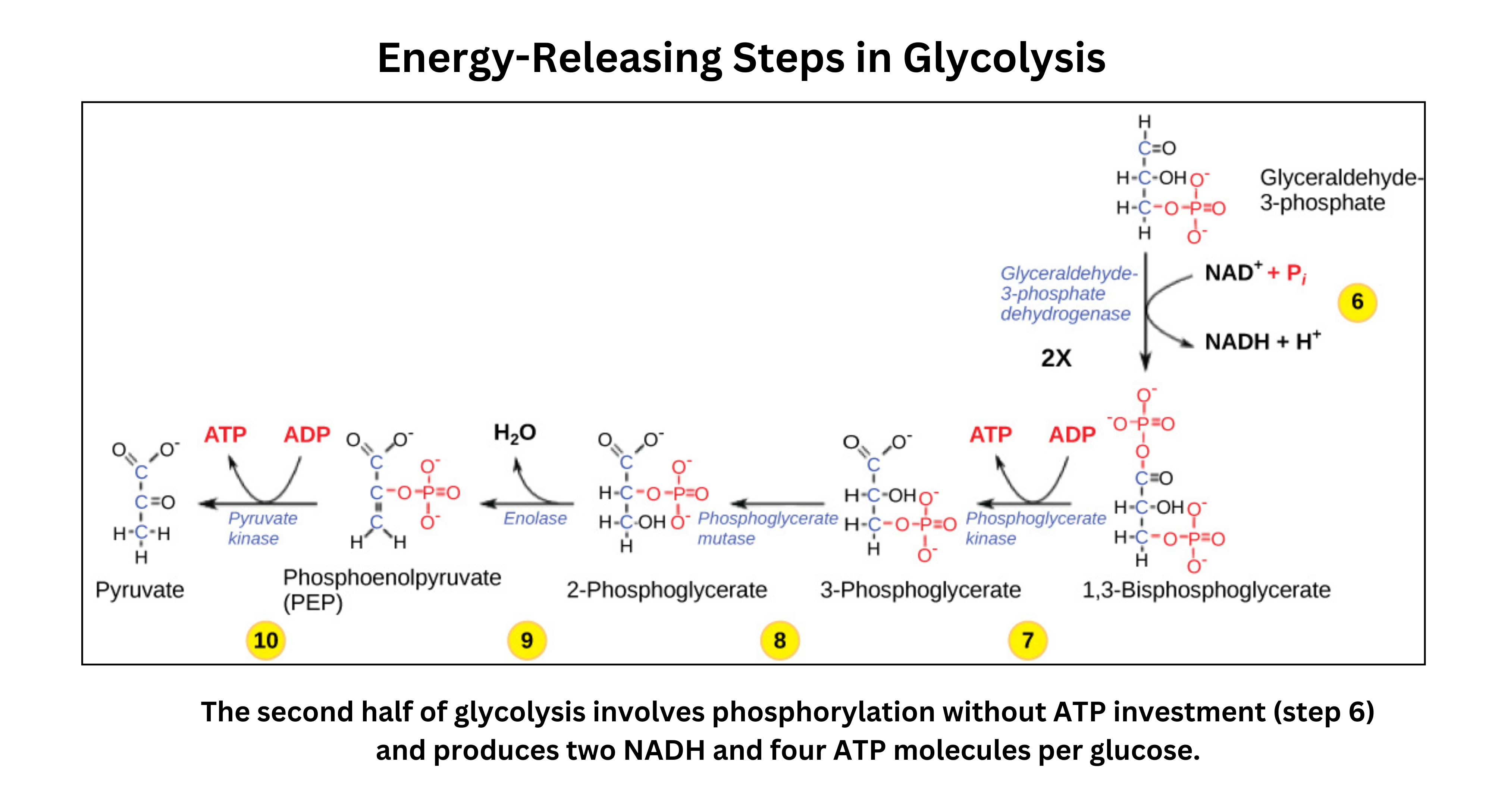
Each G3P is oxidized (producing NADH) and phosphorylated to form 1,3-bisphosphoglycerate. This phosphorylation does not require an additional ATP. - First ATP Generation
1,3-bisphosphoglycerate transfers a high-energy phosphate to ADP, producing ATP (via substrate-level phosphorylation) and 3-phosphoglycerate. - Phosphate Rearrangement
A mutase shifts the phosphate group from carbon 3 to carbon 2, forming 2-phosphoglycerate. - Dehydration
Enolase removes water, converting 2-phosphoglycerate into phosphoenolpyruvate (PEP). This step increases the phosphate group’s transfer potential. - Second ATP Formation
Pyruvate kinase transfers the phosphate from PEP to ADP, producing another ATP and forming pyruvate. Pyruvate can then be processed aerobically (in mitochondria) or anaerobically (via fermentation).
Anaerobic metabolism
Glycolysis can also function as the endpoint for anaerobic metabolism, also called fermentation. Fermentation begins with the partial oxidation of glucose to pyruvate, producing 2 net ATP per glucose. Pyruvate is then converted to regenerate NAD⁺, which is required to keep glycolysis running:
- In animals, pyruvate is reduced to lactate (lactic acid fermentation).
- In yeast, pyruvate is converted to ethanol and carbon dioxide (alcohol fermentation).
Carbohydrates stored as glycogen in animals or starch in plants can be rapidly mobilized through feeder pathways, supplying additional glucose for glycolysis.
Gluconeogenesis
Gluconeogenesis is essentially the reverse of glycolysis in overall direction: it generates glucose from non-carbohydrate precursors such as lactate, amino acids, and glycerol. This pathway is especially important during fasting or intense exercise, when certain tissues (such as the brain) require a steady glucose supply.
The pathway begins with the conversion of pyruvate to oxaloacetate by pyruvate carboxylase. This step requires ATP and uses biotin as a cofactor. Oxaloacetate is then decarboxylated and phosphorylated to form phosphoenolpyruvate (PEP) by phosphoenolpyruvate carboxykinase (PEPCK), using GTP as an energy source.
From there, subsequent reactions reverse most glycolytic steps through a series of enzyme-catalyzed reactions until fructose-1,6-bisphosphate is formed. This intermediate is then hydrolyzed by fructose-1,6-bisphosphatase, a key regulatory enzyme in gluconeogenesis. Hydrolysis removes a phosphate group to yield fructose-6-phosphate.
The pathway continues until glucose-6-phosphate is produced. Glucose-6-phosphatase then dephosphorylates glucose-6-phosphate to generate free glucose, which can be released into the bloodstream.
Gluconeogenesis is regulated at both the transcriptional and allosteric levels. Hormones such as glucagon and cortisol upregulate the pathway during low blood sugar, while insulin inhibits it when glucose is abundant.
The pentose phosphate pathway (PPP) runs parallel to glycolysis and has two key functions:
- it produces NADPH, which is crucial for reductive biosynthesis and maintaining cellular redox balance
- it generates ribose-5-phosphate for nucleotide synthesis
By diverting glucose-6-phosphate away from glycolysis, the PPP supplies both reducing power (NADPH) and building blocks needed for DNA, RNA, and other biosynthetic processes.
The net molecular and energetic outcomes of these respiration processes are coordinated through metabolic regulation. Cells maintain a dynamic steady state by controlling pathway flux through mechanisms such as allosteric regulation, hormonal signaling, and genetic control. For example, key enzymes in glycolysis and gluconeogenesis respond to cellular energy levels so that when one pathway is active, the other is suppressed. Similarly, glycogen metabolism is tuned by enzymes that control both synthesis and breakdown, allowing organisms to store energy when it’s abundant and mobilize it when needed.
Principles of metabolic regulation
Metabolic pathways are often controlled at rate-limiting steps. These are typically the slowest steps and/or the earliest irreversible reactions, making them effective control points. Regulation can be:
- Positive (amplifying a process)
- Negative (suppressing it)
These controls often form feedback loops that help maintain a dynamic steady state (homeostasis).
Glycolysis converts glucose into pyruvate and is controlled by key enzymes including hexokinase, phosphofructokinase, and pyruvate kinase. When ATP is abundant, glycolysis slows and gluconeogenesis is favored. When ADP is high (and ATP is low), glycolysis speeds up. Hormones such as epinephrine stimulate muscle glycolysis and increase blood glucose. Fructose-2,6-bisphosphate promotes glycolysis and downregulates gluconeogenesis.
Glycogen metabolism adds another layer of regulation. Glycogen breakdown produces glucose-1-phosphate, which can be converted to glucose-6-phosphate and then directed into pathways such as glycolysis or the pentose phosphate pathway. Hormones such as glucagon and epinephrine can trigger a cAMP cascade that increases glycogen phosphorylase activity (glycogen breakdown) and inhibits glycogen synthase (glycogen synthesis). When cAMP levels fall, the balance shifts toward glycogen synthesis.