Bioenergetics, free energy, ATP and redox in biology
Principles of bioenergetics
Bioenergetics concerns the flow and transformation of energy in biochemical systems.
Thermodynamics underpins this study, describing how enthalpy (), entropy (), and free energy () interact to determine whether and how a reaction proceeds.
Free energy, equilibrium, and
Concentration and Le Chatelier’s Principle
Changing reactant or product concentrations can push a reaction in one direction or the other. According to Le Chatelier’s Principle, a system shifts to counteract an imposed change and move back toward equilibrium.
Endothermic and exothermic reactions
Enthalpy () represents the heat content of a reaction. By convention,
- Endothermic processes absorb heat ().
- Exothermic processes release heat ().
Standard heats of reaction () or formation () describe enthalpy changes under standard conditions (1 bar pressure, specified temperature).
is the change in heat content during a reaction. A positive value means heat is absorbed, and a negative value means heat is released.
The standard heat of reaction () measures this enthalpy change for a specific chemical reaction under standard conditions.
Similarly, the standard heat of formation () quantifies the enthalpy change when a compound is formed from its elements in their most stable forms.
These elements are in their standard state (the lowest-energy configuration found naturally). For example, oxygen exists as (a diatomic gas) and carbon as solid graphite.
In a formation reaction, a substance is generated from its elements in their standard states (e.g., diatomic , graphite carbon).
Enthalpy is typically measured in joules () or, more commonly in chemistry, joules per mole ().
Spontaneous reactions and standard free energy change
Spontaneous reactions occur without the need for continuous external energy input. Thermodynamically, spontaneity is indicated by a negative change in free energy ().
A negative means a reaction is thermodynamically favorable, but the rate at which it proceeds depends on kinetic factors. As a result, even spontaneous reactions can be slow.
The relationship between heat exchange and spontaneity can be subtle:
- An exothermic reaction () might not be spontaneous if it is accompanied by a significant decrease in entropy ().
- An endothermic reaction () can be spontaneous if it involves a large increase in entropy.
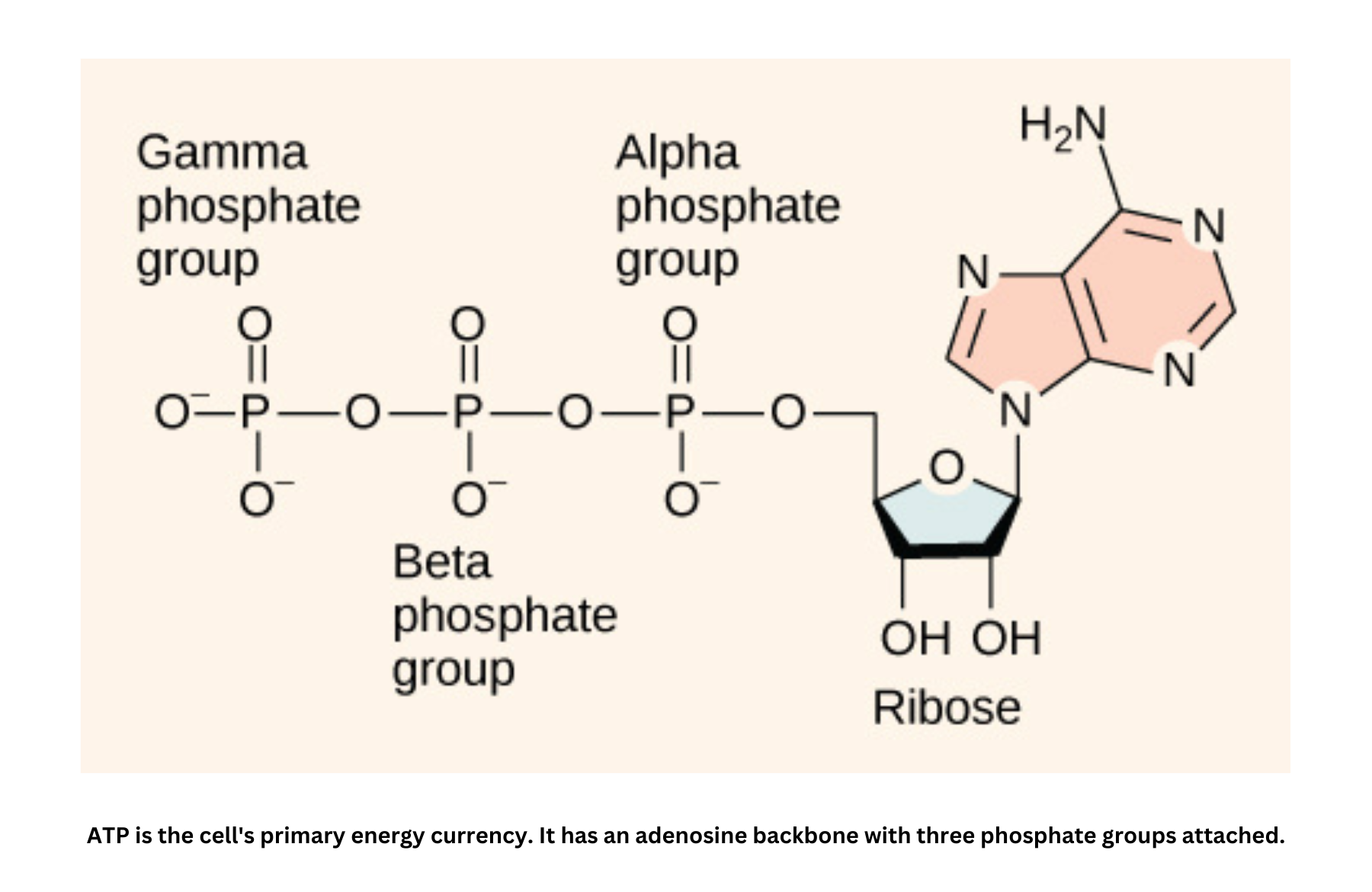
Phosphoryl group transfers and ATP
In biological systems, energy transfer is often driven by the hydrolysis of ATP, a reaction with a very negative (). The energy released can be coupled to processes such as phosphoryl group transfers.
ATP hydrolyzes into ADP in the following reaction:
Biological oxidation-reduction
Cellular energy production relies on oxidation-reduction (redox) reactions, which involve the transfer of electrons through half-reactions. These redox processes are mediated by soluble electron carriers and enzymes like flavoproteins, which facilitate electron flow and play critical roles in the cell’s bioenergetic pathways.