Carbohydrates
Carbohydrates are essential biomolecules that act as energy sources and structural components in living organisms.
They’re classified by how many sugar units they contain:
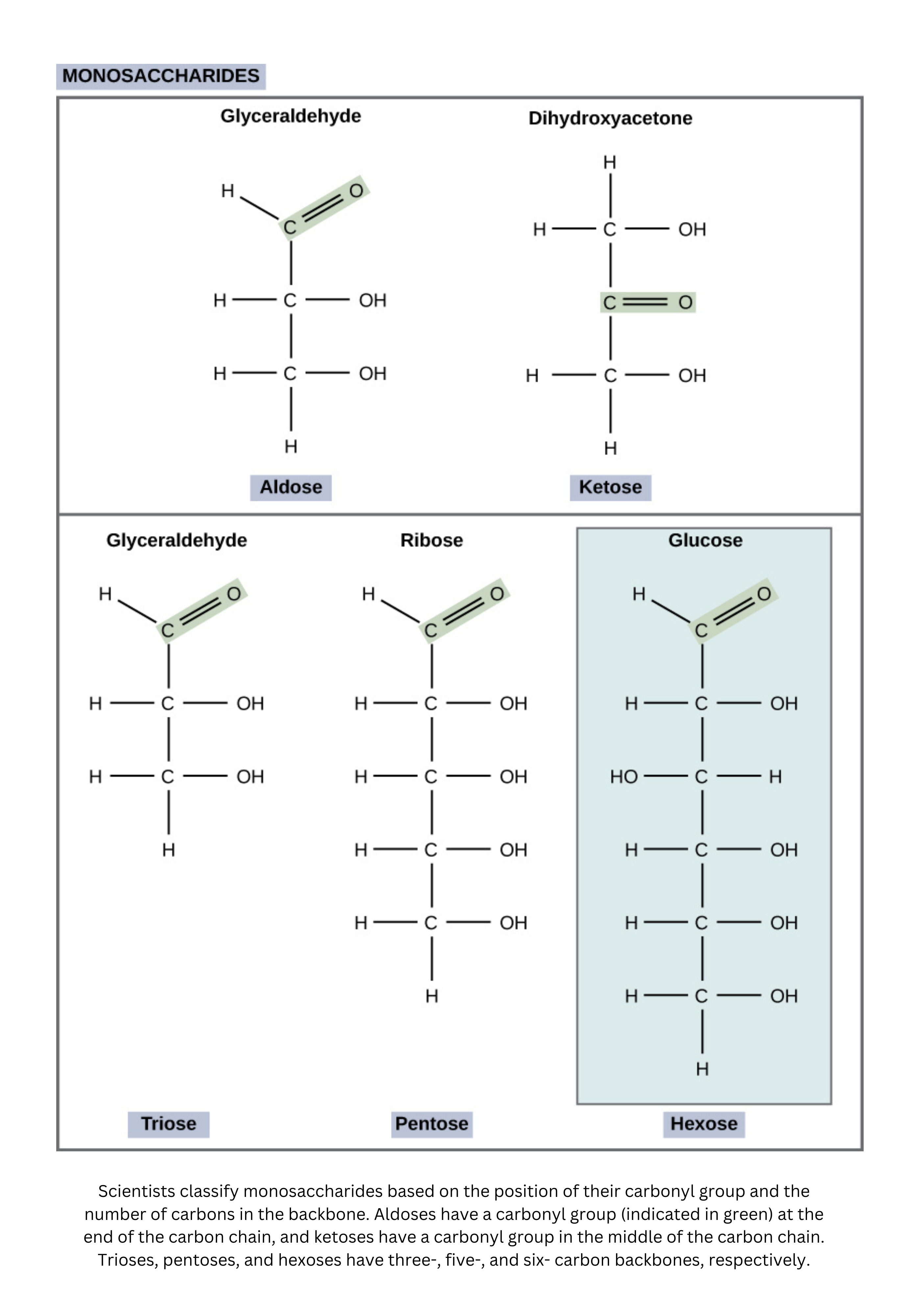
- Monosaccharides are single sugar molecules.
- Disaccharides consist of two linked sugars.
- Polysaccharides are long chains of sugars.
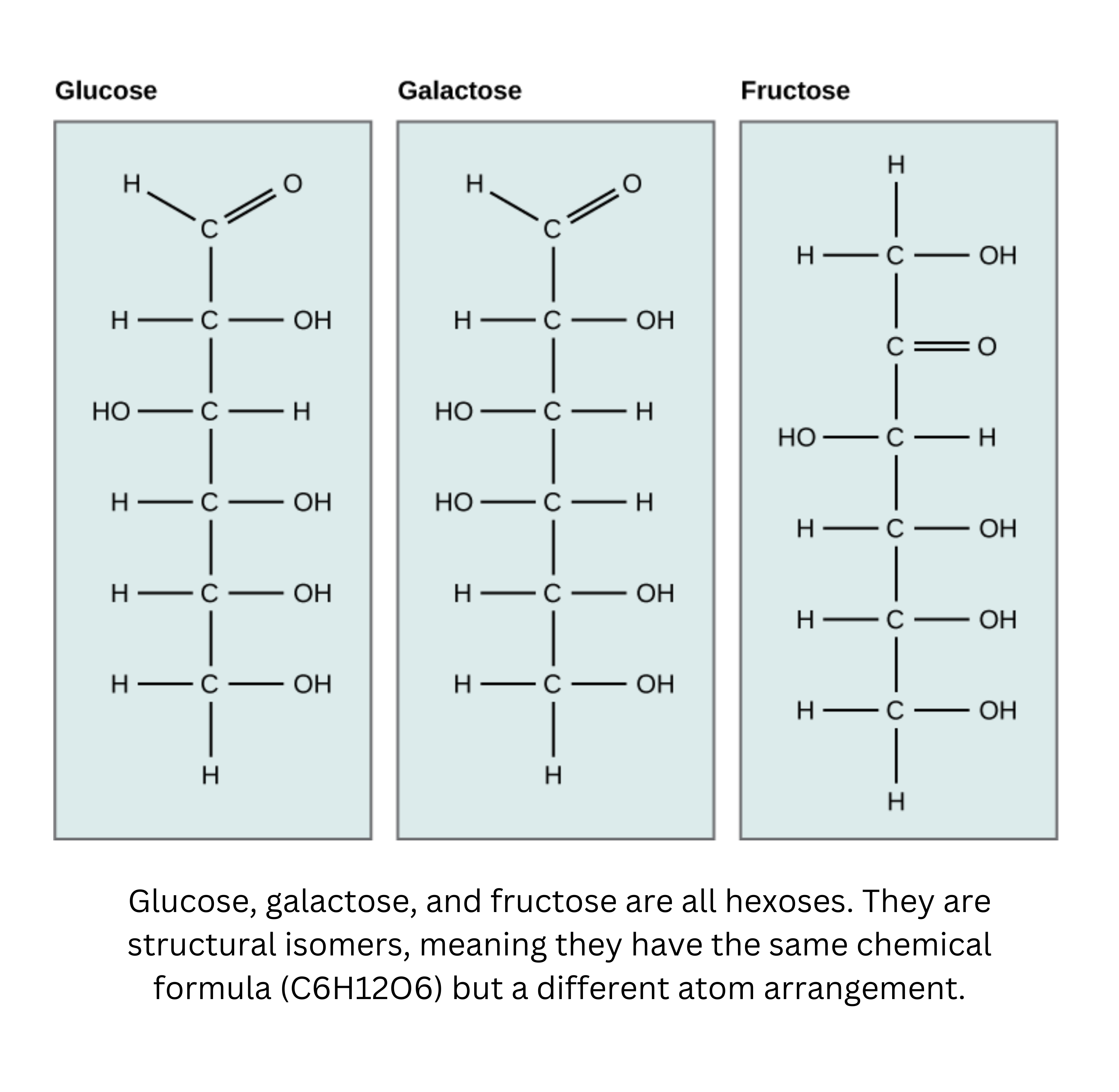
Glucose, with the chemical formula , is a major energy source for humans. Other important monosaccharides include galactose (a component of lactose, or milk sugar) and fructose (found in sucrose, the sugar in many fruits and in table sugar).
Even though glucose, galactose, and fructose share the same molecular formula (), they’re structural isomers. That means they differ in how their atoms are connected and arranged. Their different chemical behaviors come from differences in the placement of functional groups around their asymmetric carbons. Importantly, each of these sugars has more than one chiral center.
Nomenclature
- Carbohydrates can be named using both systematic IUPAC nomenclature and common names, and their classification points to their diverse functions in biology.
Carbohydrate prefixes:
- Deoxy = “without oxygen” has an in place of an at a certain position.
- D/L = absolute configuration = the prefixes D and L indicate the absolute configuration of a sugar. This designation is based on the stereochemistry (3D arrangement of atoms) of the chiral carbon farthest from the carbonyl group. If the hydroxyl group on this carbon is oriented like it is in D-glyceraldehyde, the sugar is designated D; the opposite configuration is L.
- / = anomeric configuration= arises when a sugar forms a cyclic structure. During cyclization, the carbonyl group reacts with a hydroxyl group to create a new chiral center called the anomeric carbon.
- The form has the hydroxyl group on the anomeric carbon positioned opposite to the CH₂OH group at the far end of the ring.
- The form has the hydroxyl group on the same side as the group.
- These configuration differences matter because they change a sugar’s physical and chemical properties and can affect biological interactions.
Carbohydrate suffix: all sugars end in -ose.
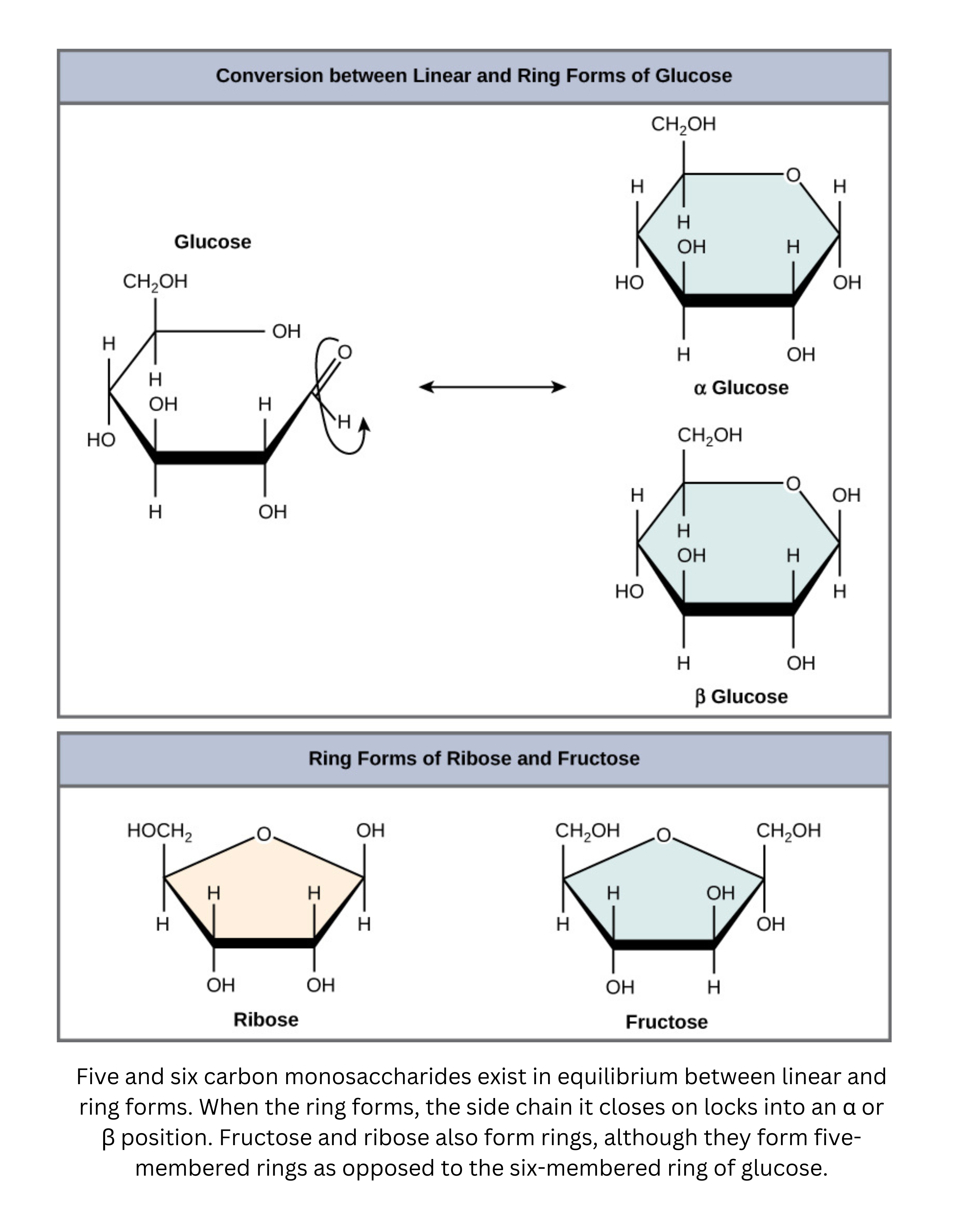
Ring (cyclic) forms of monosaccharides
- Many hexoses, such as glucose, exist mainly in cyclic forms rather than as open-chain molecules, especially in aqueous solution. Ring formation happens through an intramolecular reaction that produces either a hemiacetal (an alcohol and ether attached to the same carbon) or a hemiketal (formed when an alcohol adds to a ketone). These cyclic sugars can adopt characteristic conformations (such as chair or boat forms), which influence stability and reactivity.
Within cyclic forms, small stereochemical differences produce epimers and anomers.
- Epimers differ in configuration at just one specific carbon atom.
- Anomers are a special type of epimer that differ at the new chiral center created during cyclization (the anomeric carbon). These are labeled alpha () or beta () based on the orientation of the substituent relative to the ring.
Disaccharides and polysaccharides
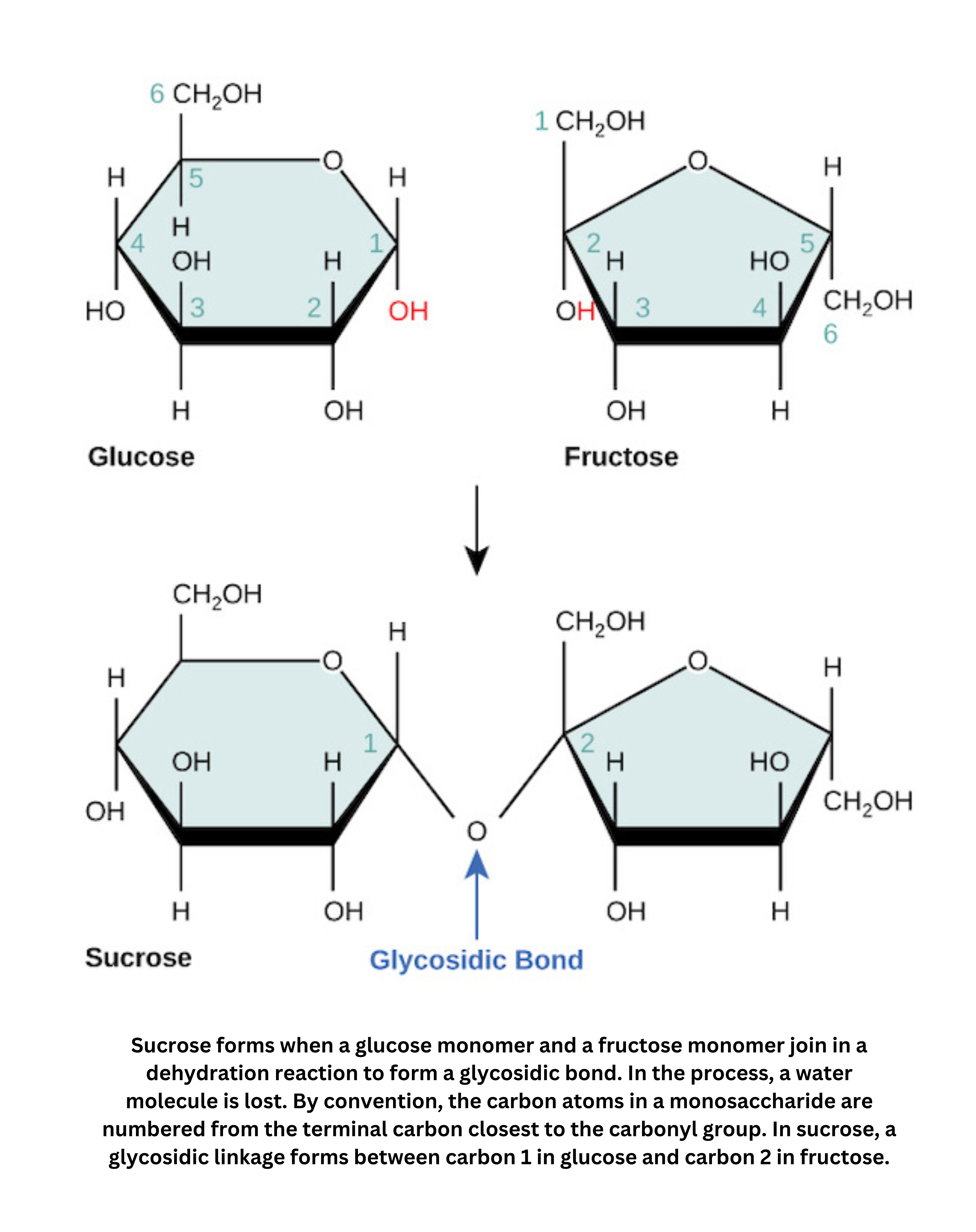
Glycosidic bonds join monosaccharides into larger carbohydrate molecules.
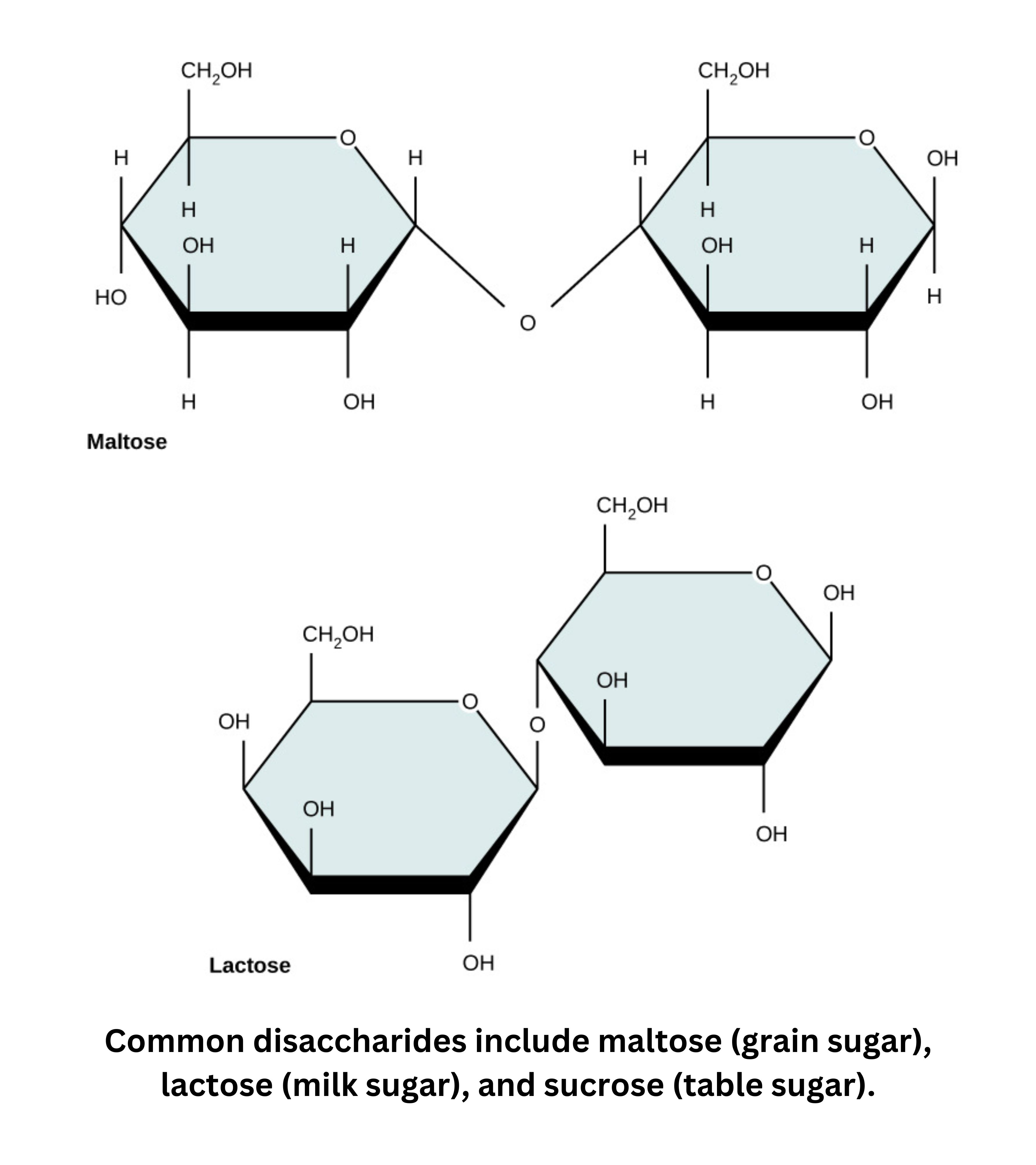
- Sucrose (table sugar) is a disaccharide made of -glucose and -fructose. They’re connected through their anomeric hydroxyl groups, forming acetal bonds.
- Lactose forms when -galactose links to glucose (which can be either or ) through a 1-4 glycosidic bond.
- Maltose (malt sugar) is another disaccharide. It forms through a Dehydration reaction between two glucose molecules.
Starch is a polymer (a polysaccharide made up of monosaccharides) composed of glucose units connected mainly by 1-4 glycosidic bonds. It serves as the main energy storage molecule in plants.
Glycogen is structurally similar to starch but includes additional 1-6 linkages that create branching. This branching makes glycogen an efficient energy reserve in animals, and it’s stored primarily in the liver.
Reactions of monosaccharides
- Acetal formation occurs when an extra hydroxyl group attacks a carbonyl carbon. This reaction can link monosaccharides into polysaccharides if the attacking - comes from another sugar unit.
- Mutarotation is the equilibrium between the and forms (anomers) of a sugar.
- Under strong oxidation conditions, the aldehyde group and terminal hydroxyls are converted into carboxylic acids, while other hydroxyl groups are transformed into ketones. In extreme cases, such as during cellular respiration, all carbon atoms can be fully oxidized to .
- In contrast, mild oxidation is more selective. For example, Tollens reagent oxidizes the aldehyde group of aldoses to a carboxylic acid, and nitric acid oxidizes both the aldehyde and the terminal hydroxyl groups to carboxylic acids while leaving the other hydroxyl groups intact.
- Reduction reactions convert monosaccharides into polyalcohols by turning carbonyl groups into additional hydroxyl groups.
Hydrolysis of the glycoside linkage
- Another critical reaction in carbohydrate chemistry is the hydrolysis of the glycosidic linkage.
- Hydrolysis, often catalyzed by glycosidase enzymes, breaks these bonds by adding a water molecule, releasing the constituent sugar units.