Light, electromagnetic radiation
Interference; Young’s double-slit experiment
Light is a form of electromagnetic radiation that can behave like a wave. One key wave behavior is interference, where waves overlap and combine.
A classic demonstration is Young’s double-slit experiment. Light passes through two parallel slits, and the waves from the two slits overlap on a screen.
For a stable interference pattern, the light from the two slits must be:
- Coherent, meaning the waves maintain a constant phase relationship.
- Monochromatic, meaning the light has a single wavelength.
The locations of the bright fringes are described by
where is the distance between the slits, is the angle of the fringe relative to the center, is the order of the fringe, and is the wavelength.
- Bright fringes appear at , etc.
- Dark fringes occur at , etc.
Thin films, diffraction grating, single-slit diffraction
Diffraction is another wave behavior of light. It occurs when light spreads out after passing through an opening or around an obstacle, which can then lead to interference patterns.
In thin films, light reflects from both the outer and inner surfaces of the film. The two reflected waves can interfere with each other, producing colorful patterns (for example, an oil film on water).
Other diffraction phenomena, X-ray diffraction
Diffraction shows up whenever light interacts with small openings or edges. Instead of forming a perfectly sharp image or shadow, the light produces alternating bright and dark regions due to interference.
For example, when light shines through a pinhole, the screen shows concentric bright and dark circles, with a prominent bright spot at the center.
Similarly, when light passes the edge of an opaque boundary, the shadow edge is not perfectly sharp. Fringes of alternating brightness appear along the boundary.
Another example is light passing by a small object like a penny. The shadow is not completely dark; it can show a bright central spot surrounded by rings of light and dark. This is another clear sign of light’s wave behavior.
In X-ray diffraction, high-energy X-rays are directed at a crystal. The X-rays diffract from the regularly spaced atoms, and the resulting interference pattern (spots or rings) can be used to determine details of the crystal’s molecular structure.
Polarization of light: linear and circular
Polarization describes the direction of oscillation of light’s electric field.
- In unpolarized light, the electric field vibrates in many directions.
- In polarized light, the electric field is restricted to a particular direction (for example, a single plane for linear polarization).
This property can be produced or used in several ways:
- Selective absorption uses a polarizer to block all but the light waves that vibrate in one chosen direction.
- Reflection at specific angles can polarize light, with the reflected waves aligned in a single plane.
- Double refraction occurs in birefringent materials, which split an incoming beam into two rays with perpendicular polarizations.
- Scattering by air molecules produces partially polarized light, and optically active substances can rotate the plane of polarization clockwise or counterclockwise.
The Doppler effect describes how the frequency of light changes when there is relative motion between the light source and the observer.
- If the source and observer move apart, the observed frequency decreases, producing a red shift.
- If they move toward each other, the observed frequency increases, producing a blue shift.
The equations used for these shifts are analogous to those for sound waves, except that the speed of light replaces the speed of sound.
The Doppler effect equation
…where:
- is the frequency experienced by the observer
- is the frequency emitted by the source
- is the speed of the wave (most frequently the speed of sound or light)
- is the velocity of the observer ( if it is at rest)
- is the velocity of the source ( if it is at rest)
| Doppler shift | Stationary observer | Observer moving towards source | Observer moving away from source |
|---|---|---|---|
| Stationary source | |||
| Source moving towards observer | |||
| Source moving away from observer |
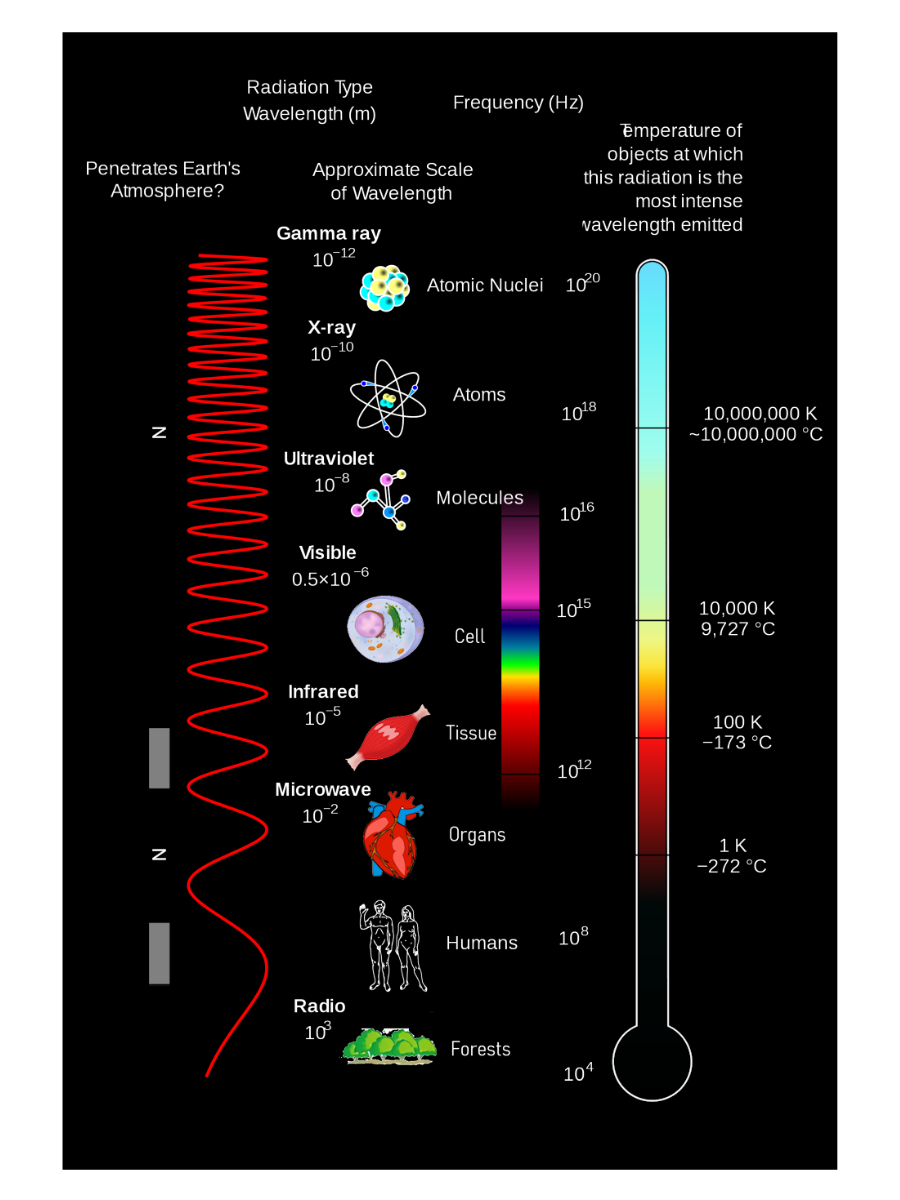
Classification of electromagnetic spectrum, photon energy
The photon model of light treats electromagnetic radiation as packets of energy called photons. Each photon’s energy is proportional to the radiation’s frequency:
,
where is Planck’s constant and is frequency.
Because wavelength and frequency are related by the speed of light
$ c = \lambda f $,
you can also write photon energy as .
The electromagnetic spectrum covers many types of radiation, distinguished by wavelength and energy:
- Radio waves have the lowest energy and longest wavelengths.
- Microwaves have slightly higher energy, useful for radar and cooking.
- Infrared light (IR) has moderate energy and is used in remote controls and night vision.
- Visible light lies in the middle of the spectrum and is responsible for human color vision.
- Ultraviolet (UV) light, X-rays, and gamma rays have progressively higher energy and can be ionizing - able to eject electrons from molecules and potentially cause health hazards such as cancer.
Properties of electromagnetic radiation
Electromagnetic radiation travels at a maximum speed of (about ) in a vacuum.
In a material medium, the speed is lower. The index of refraction relates the two speeds:
,
where is the index of refraction, is the radiation’s speed in the medium, and is the speed of light in a vacuum.
As an electromagnetic wave, it consists of oscillating electric and magnetic fields. These fields are perpendicular to each other and to the direction the wave travels.
The electromagnetic spectrum runs from low-frequency, long-wavelength radiation to high-frequency, short-wavelength radiation.
- At the low-frequency end are radio waves, which can drive oscillations of electrons in antennas.
- Microwaves are higher in frequency and can induce molecular rotation.
- Infrared waves can cause molecular vibration.
Visible light can excite electrons within atoms and spans roughly 400 nm (violet) to 700 nm (red).
Ultraviolet waves can carry enough energy to break bonds or eject electrons and are often classified as ionizing radiation. X-rays are even more energetic, also ionize matter, and can trigger the photoelectric effect.
At the highest frequencies are gamma rays, which have even greater energy and penetrating power than X-rays.
Visual spectrum, color
The visual spectrum is the range of wavelengths and frequencies that the human eye detects as different colors.
- Blue light has a shorter wavelength and higher frequency, so each photon carries more energy (, where is Planck’s constant and is frequency).
- Red light has a longer wavelength and lower frequency, so each photon carries less energy.
A laser (Light Amplification by Stimulated Emission of Radiation) produces light by stimulated emission, rather than the spontaneous emission typical of ordinary light. Inside the lasing medium, light is repeatedly amplified as it reflects back and forth, producing a coherent, highly focused beam.