Gas phase
Absolute temperature, () Kelvin scale
The absolute temperature scale is measured in Kelvin (). Its zero point is absolute zero, the lowest possible temperature, where no more thermal energy can be removed from a substance.
The Kelvin scale lines up with the Celsius () scale, using the relationship:
So, water freezes at () and boils at ().
The Fahrenheit () scale is still widely used in some regions, where:
The Kelvin scale is especially important in science because it starts at the theoretical point of zero thermal energy.
| Description | Celsius () | Kelvin () | Fahrenheit () |
|---|---|---|---|
| Surface of the sun | 5600 | 5900 | 10100 |
| Boiling point - water | 100 | 373 | 212 |
| Body temperature | 37 | 310.2 | 98.6 |
| Room temperature | 20 | 293 | 68 |
| Cool day | 10 | 283 | 50 |
| Freezing point - water | 0 | 273 | 32 |
Pressure, simple mercury barometer
Pressure is the force exerted per unit area. It’s defined by:
where is the applied force and is the area over which the force acts.
At sea level, atmospheric pressure is about , which is equivalent to , and it decreases with increasing elevation. A mercury barometer measures atmospheric pressure by letting the atmosphere push a column of mercury upward. One end of the barometer is open to the air, while the other end is sealed, forming a vacuum.
- The pressure is the weight of the mercury that is lifted () divided by the cross-sectional area () of the tube.
- Standard barometers are calibrated so that raises the mercury to , also written as or .
- When using the formula, make sure force is in Newtons () and area is in square meters (). This gives pressure in Pascals ().
Molar volume
The molar volume at 0 degrees Celsius and is . In other words, ideal gases occupy liters per mole of molecules under these standard conditions. (It’s per mole, not moles per liter.)
A mole contains about particles, which is an enormous number of molecules and can take up a substantial volume. For example, air is mostly nitrogen; in its diatomic form (), nitrogen has a molar mass of about grams. Because air is light, a full mole of it occupies liters at standard conditions.
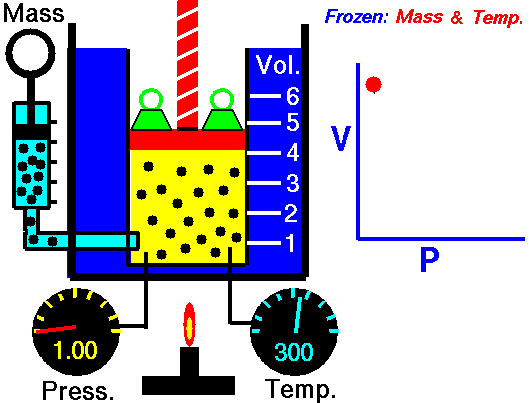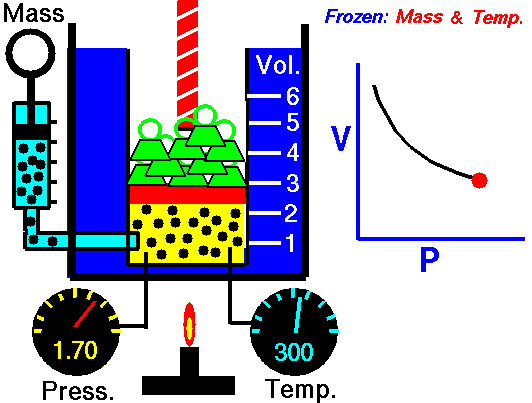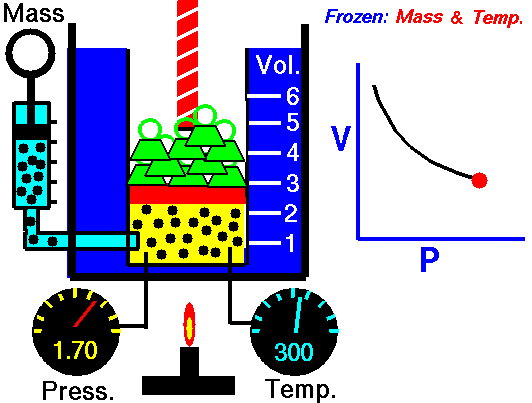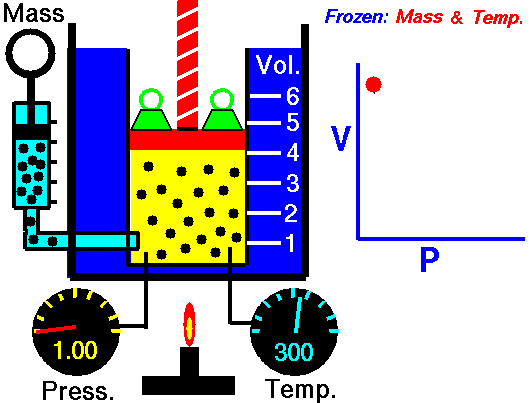
Ideal gas
An ideal gas is described by the kinetic molecular theory, which models a gas as a collection of point particles that move randomly and collide elastically with each other and with the container walls.
In an ideal gas, the molecular volume is negligible and there are no intermolecular forces. This behavior is a good approximation at low pressure and high temperature, where molecules are far apart. At high pressures and low temperatures, molecules are closer together, so intermolecular interactions and finite molecular volume become important, and the gas can eventually condense.
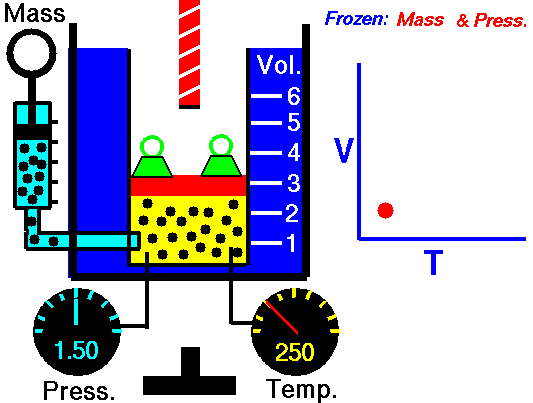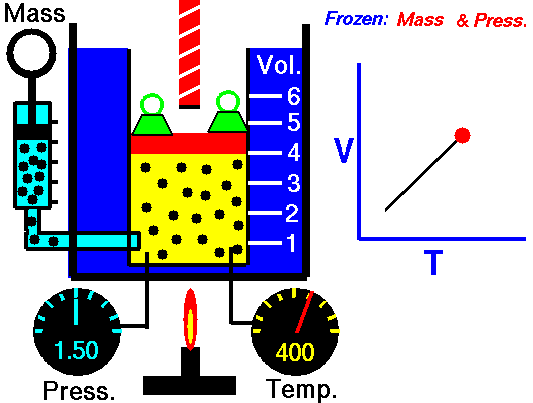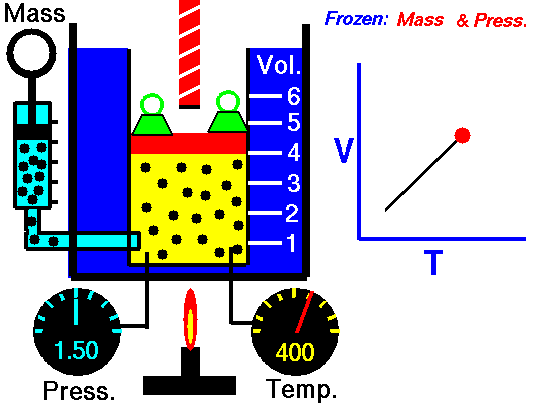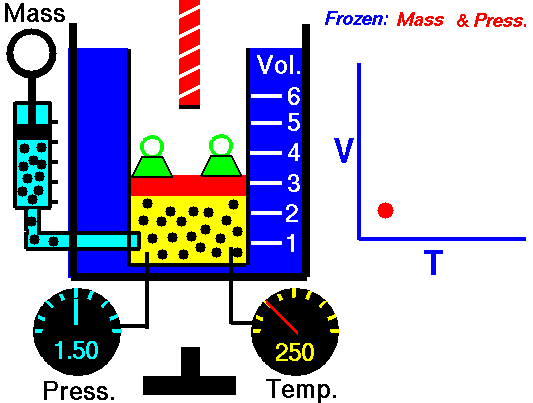
This law leads to the combined gas law: when and are constant, the ratio stays constant. From this relationship, you can derive:
- Boyle’s law: at constant
- Charles’s law: at constant
- Avogadro’s law: equal volumes of gas at the same temperature and pressure contain equal numbers of moles
Kinetic molecular theory of gases
The kinetic molecular theory explains the behavior of gases and supports the ideal gas laws. It assumes that gas molecules:
- are in constant, random molecular motion
- experience no intermolecular forces
- have negligible molecular volume
- undergo perfectly elastic collisions, so the total kinetic energy stays constant during collisions
In this model, pressure comes from molecules colliding with the container walls. Because collisions occur randomly in all directions, the pressure is uniform throughout the container. Temperature measures the average kinetic energy of the gas molecules, so:
- higher temperatures correspond to faster molecular speeds
- lower temperatures correspond to slower molecular motion
Heat capacity at constant volume and at constant pressure
The heat capacity at constant volume is the heat required to raise a system’s temperature by one degree while keeping volume fixed. Because the volume doesn’t change, the system does no expansion work.
The heat capacity at constant pressure is the heat needed to raise the temperature by one degree while keeping pressure constant. Here, the system can expand, so some energy goes into expansion work. That’s why the constant-pressure heat capacity is higher than the constant-volume heat capacity. The difference between the two is explained by the work done against the surroundings.
Boltzmann’s constant is a fundamental value that links the average kinetic energy of individual particles to the macroscopic temperature of a system. It connects microscopic particle behavior to bulk thermal properties.
Diffusion and effusion, graham’s law
Diffusion is the random movement of molecules from regions of higher concentration to regions of lower concentration (down a concentration gradient). Effusion is the escape of gas molecules through a very small opening in a container, also driven by random molecular motion.
Deviation of real gas behavior from the Ideal Gas Law, Van der Waals
In an ideal gas, molecules are treated as having negligible volume and no interactions. This works well at low pressure and high temperature, when molecules are far apart.
At higher pressures or lower temperatures, molecules are closer together and begin to experience intermolecular attractions. These attractions pull molecules toward each other and effectively reduce the measured pressure. When molecules are forced extremely close together, their finite size produces steric repulsion, which pushes them apart and increases the pressure.
These deviations from ideal behavior are described by the Van der Waals equation, which includes two constants:
- accounts for attractive forces (lowering the pressure)
- accounts for finite molecular volume (raising the pressure)
Partial pressure, mole fraction, and Dalton’s Law
Partial pressure is the pressure contributed by one gas in a mixture. The total pressure is the sum of the partial pressures of all gases present.
The fraction of the mixture made up by a particular gas is its mole fraction. It’s defined as the moles of that gas divided by the total moles of gas in the mixture.