Electrochemistry and nerve cells
Electrochemistry
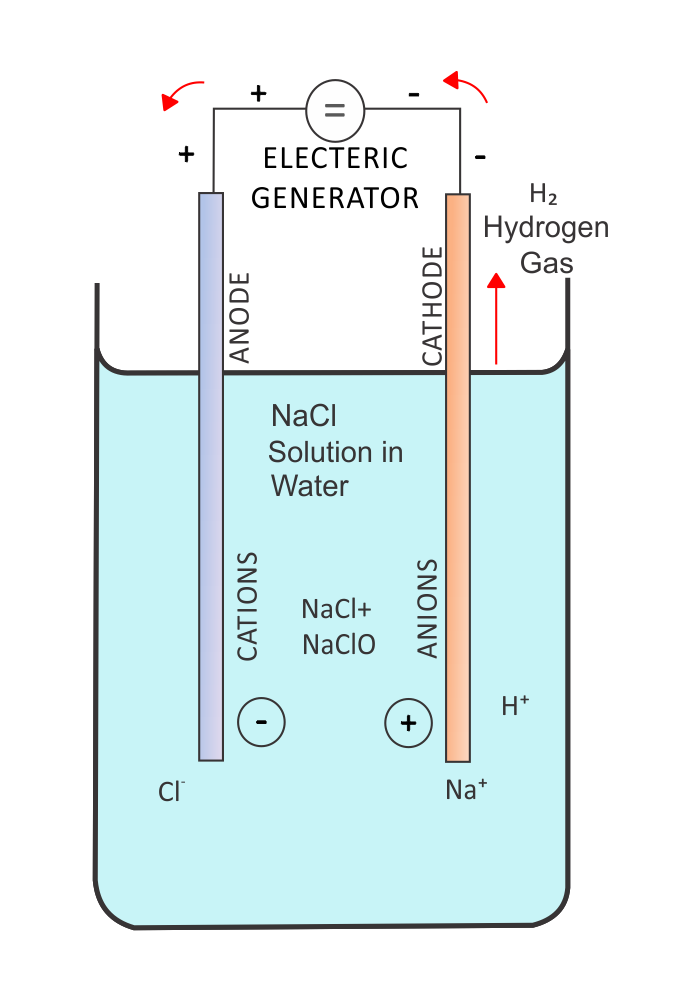
Electrolysis is a process where an external voltage source (typically a battery) drives chemical reactions that would not occur spontaneously.
In an electrolytic cell, the inherent cell potential is negative, so the applied voltage must be greater than the magnitude of this negative potential to force the reaction forward. In other words, the battery supplies enough energy to push electrons in the nonspontaneous direction.
In contrast, a galvanic cell (or voltaic cell) naturally produces a positive cell potential and does not require an external voltage source. In diagrams, red arrows often show the electron flow forced by the battery in an electrolytic cell, highlighting that the electrons are moving opposite to the spontaneous direction.
Anode, cathode, electrolyte
In both electrolytic and galvanic cells:
- The anode is where oxidation occurs (electrons are released).
- The cathode is where reduction occurs (electrons are absorbed).
These cells also require an electrolyte, a medium containing mobile ions. The ions move through the solution to balance charge as electrons move through the external wire. Without an electrolyte, charge would quickly build up and the circuit would stop.
Faraday’s law at an electrode
Faraday’s law connects how much substance is produced or consumed at an electrode to the current passing through the cell.
- Current is the rate of charge flow: .
- Faraday’s constant is the charge per mole of electrons: .
Because , we can combine these relationships to get:
This says: the total charge delivered () equals the number of moles of electrons transferred () times Faraday’s constant ().
To find how many moles of a substance are deposited (or produced), you then use the stoichiometry of the relevant half-reaction. For example, in
→
1 mole of copper is deposited for every 2 moles of electrons transferred.
Electron flow; oxidation, and reduction at the electrodes
In an electrochemical cell, electron flow is tied directly to the redox reactions at the electrodes.
- At the anode, oxidation occurs: a species loses electrons, for example .
- Those electrons travel through the external circuit to the cathode.
- At the cathode, reduction occurs: a species gains electrons, for example .
Oxidation increases a species’ charge by removing electrons, while reduction decreases charge by adding electrons.
Galvanic or voltaic cells
Half-reactions, reduction potentials, cell potential
A galvanic cell (or voltaic cell) converts chemical energy into electrical energy using a spontaneous redox reaction. The overall reaction is split into two half-reactions:
- an oxidation half-reaction (electrons are produced)
- a reduction half-reaction (electrons are consumed)
Each half-reaction has a reduction potential, which measures how strongly a species tends to gain electrons (be reduced). The oxidation potential for the reverse process is the negative of the reduction potential for the same half-reaction.
The overall cell potential is found by adding:
- the reduction potential at the cathode
- the oxidation potential at the anode
A positive cell potential means the cell operates spontaneously and can produce electric current.
For instance, if the cathode reaction is with a reduction potential of and the anode reaction is (with an oxidation potential of ), then the cell potential is , confirming the cell’s spontaneous operation.
Direction of electron flow
In any electrochemical system, electrons flow from the anode to the cathode.
- At the anode, oxidation produces electrons (and typically produces cations).
- Those electrons move through the external circuit to the cathode, where reduction consumes them.
A quick reminder is “A to C”: anode to cathode.
In galvanic cells, the electrode roles arise naturally: the species with the highest oxidation potential (or lowest reduction potential) serves as the anode, and the species with the highest reduction potential serves as the cathode. This arrangement gives spontaneous electron flow.
In electrolytic cells, an external battery forces electron flow, even when that means driving a nonspontaneous reaction. In both types of cells, the circuit is completed by electrons moving through the wire and ions moving through the electrolyte to maintain charge balance.
Concentration cell
A concentration cell is made by connecting two nearly identical half-cells that use the same half-reaction and identical electrodes. The only difference is the concentration of one redox species, so the cell’s potential comes entirely from that concentration difference.
Batteries
A battery is a specially designed galvanic cell used to provide electrical power for specific applications. A battery provides an emf (electromechanical force) that ideally equals the terminal voltage if there is no internal resistance. In real batteries, internal resistance causes a voltage drop, so the terminal voltage is lower than the emf.
Some common battery types:
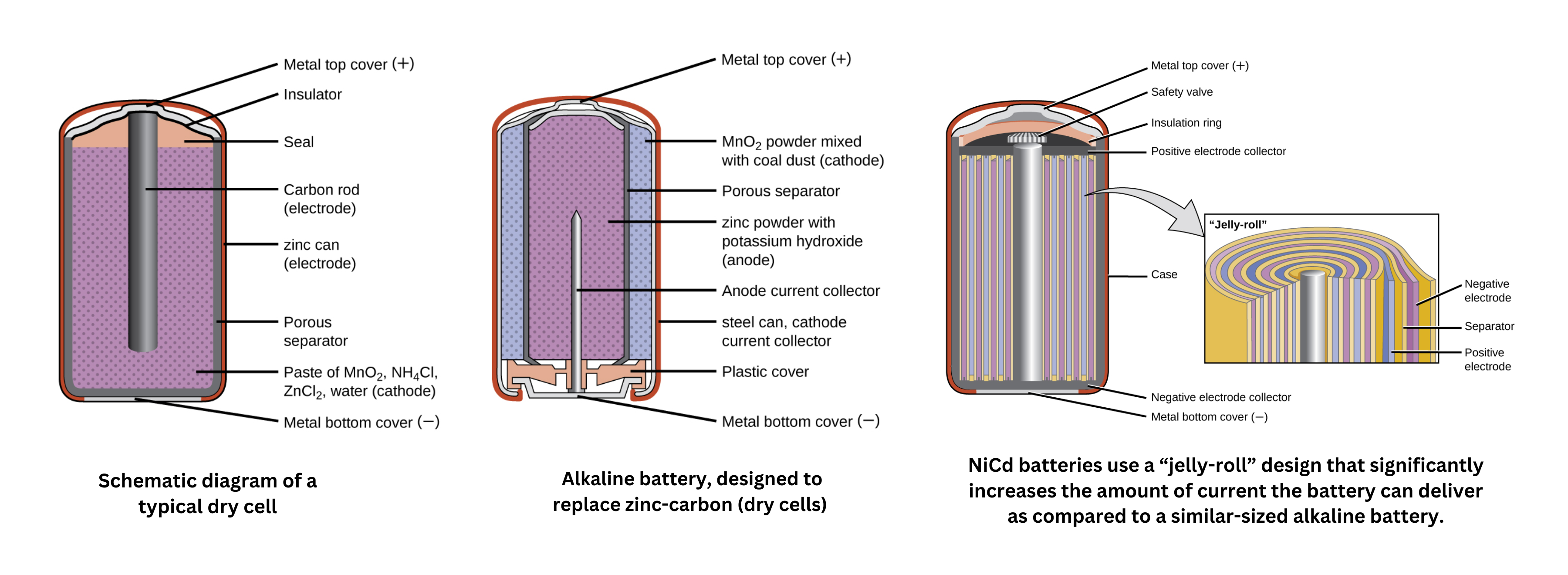
- The dry cell is one common type, using a zinc can as both the container and the anode (negative terminal) and a graphite rod as the cathode (positive terminal). The zinc can is filled with an electrolyte paste - composed of manganese(IV) oxide, zinc(II) chloride, ammonium chloride, and water - that facilitates the redox reactions, with the graphite rod immersed to complete the cell.
- Alkaline batteries build on this design by employing an alkaline electrolyte, typically potassium hydroxide, to improve performance while still using similar redox couples.
- The nickel-cadmium () battery is a rechargeable option, consisting of a nickel-plated cathode and a cadmium-plated anode with a potassium hydroxide electrolyte. In these batteries, the positive and negative plates are kept separate by a separator to prevent short circuits, then rolled together and enclosed in a case.
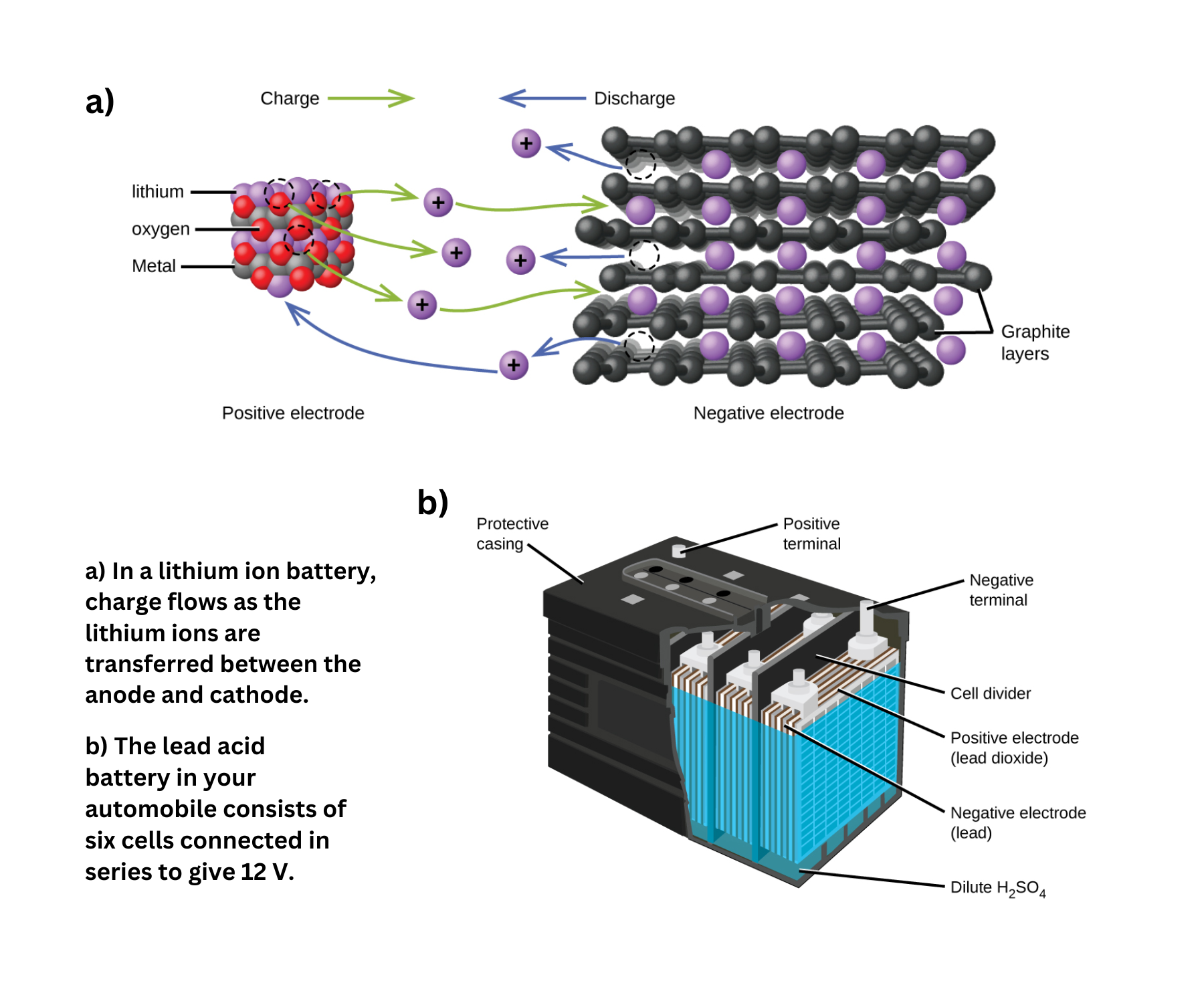
- Lithium ion batteries are widely used in portable electronics due to their ability to deliver high current, their lightweight nature, a nearly constant discharge voltage, and a low self-discharge rate.
- The lead acid battery is commonly found in automobiles because it is inexpensive and capable of generating the high current necessary to start engines.
Specialized cell - nerve cell
Attached to the cell body of nerve cells are dendrites, branching structures that serve as the neuron’s receptive region and increase surface area for incoming signals.
Extending away from the cell body is a single axon, which carries electrical impulses toward the axon terminals - sometimes called synaptic knobs or boutons - where neurotransmitters are released.
The axon may be wrapped in a myelin sheath, produced by Schwann cells in the peripheral nervous system or by oligodendrocytes in the central nervous system. This myelin sheath is made of fatty layers that act as insulation along the axon, with regular gaps called nodes of Ranvier. Because these nodes lack myelin, the action potential jumps from one node to the next, which greatly speeds up nerve impulse conduction.


-cells--20250718135943--f472808ae8327ebbff0064c0ad386b422aa0fb4c065aaecc02313908a2318a54.png)