Cell cycle and mitosis
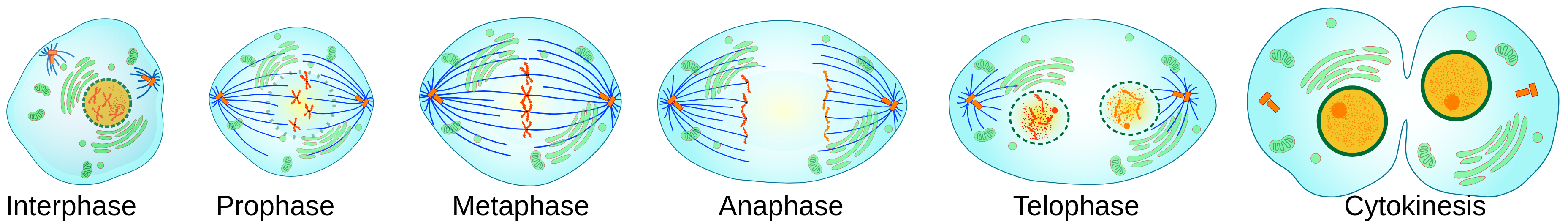
The phases of the cell cycle are , , , , and .
- In , cells are quiescent: they are not replicating their DNA and not dividing. Examples include neurons and muscle cells.
- involves cell growth and organelle production, while is the synthesis phase in which DNA and centrioles replicate.
- During , the cell continues to grow and prepares for mitosis.
- In the final phase, cells undergo mitosis.
- Interphase combines , , and . This is a period of intense metabolic activity and preparation for cell division.
Mitosis itself is further divided into:
- Prophase: Chromatin condenses into distinct chromosomes, the nuclear membrane disassembles, and the mitotic spindle begins forming, with centriole pairs migrating to opposite poles.
- Metaphase: Chromosomes align at the cell’s midline; spindle fibers attach to kinetochores located at each chromosome’s centromere.
- Anaphase: Sister chromatids separate and are pulled toward opposite poles by the spindle apparatus.
- Telophase: Chromosomes decondense, the nuclear membrane re-forms around each set of chromosomes, and the mitotic spindle disassembles, essentially reversing prophase.
Mitotic structures
During mitosis, several key structures work together:
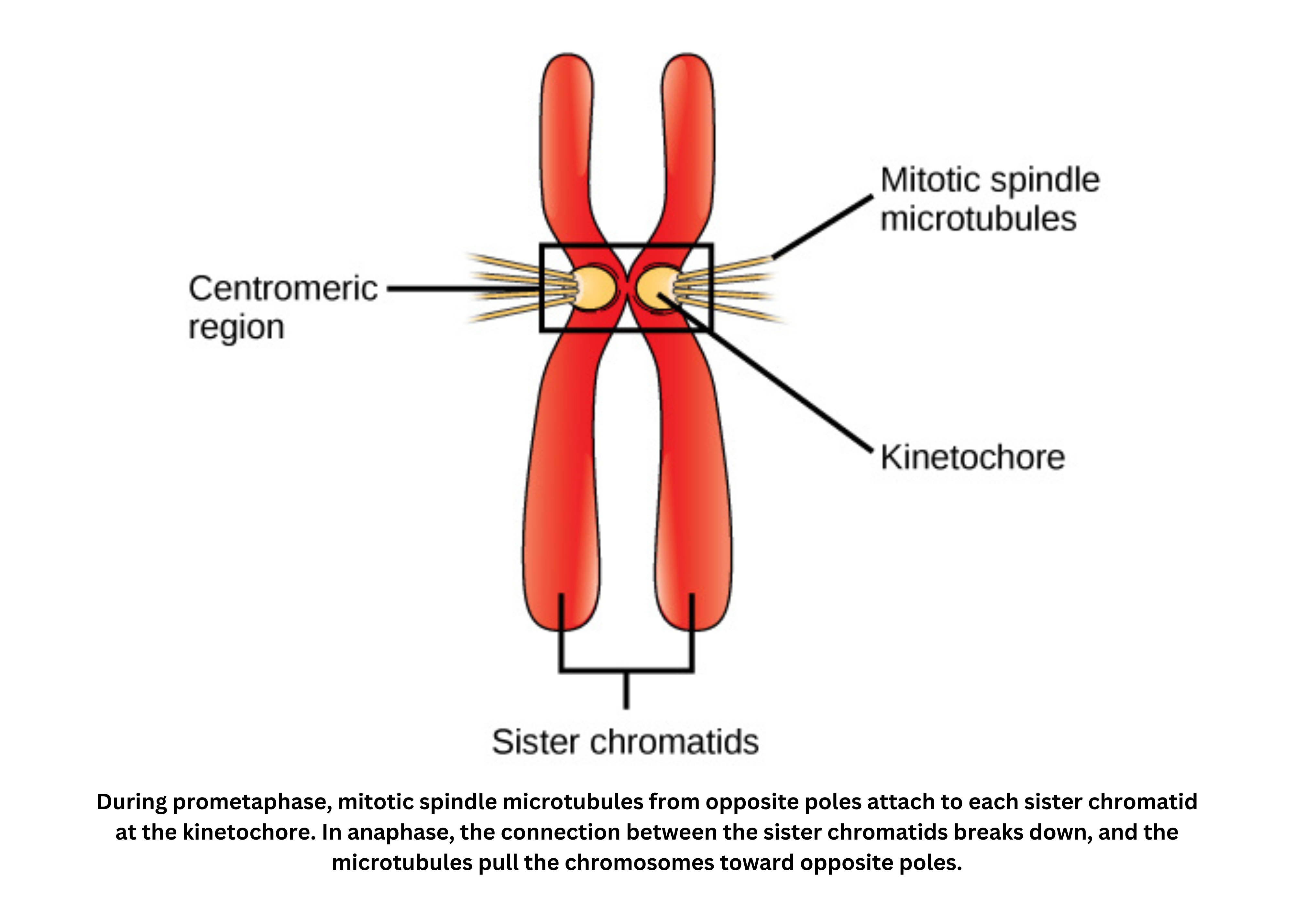
- At the cell poles, centrioles act as microtubule-organizing centers. They help form asters (radial arrays of microtubules), which contribute to building the spindle apparatus. The spindle is a network of microtubules and associated proteins that attaches to chromosomes and is responsible for pulling apart and guiding chromatids to each daughter cell.
Each chromosome is made up of two identical chromatids joined at the centromere. At the centromere, a specialized protein complex called the kinetochore forms and provides the attachment site for spindle fibers.
Early in mitosis, the nuclear membrane breaks down so the spindle can access the chromosomes. As mitosis concludes, the nuclear envelope is reassembled around the separated chromatids, restoring a nucleus in each daughter cell.
Chromosome movement is driven by the dynamic instability of microtubules and the action of motor proteins. Together, these mechanisms ensure chromatids are pulled accurately to opposite poles.
Growth arrest
Cells can stall the cell cycle (growth arrest) if there is substantial genetic damage (this can stop the cycle in the M phase). Contact inhibition occurs when epithelial cells signal that available space has been maximized (cells are touching). Growth arrest can also be triggered when nutrients are insufficient.
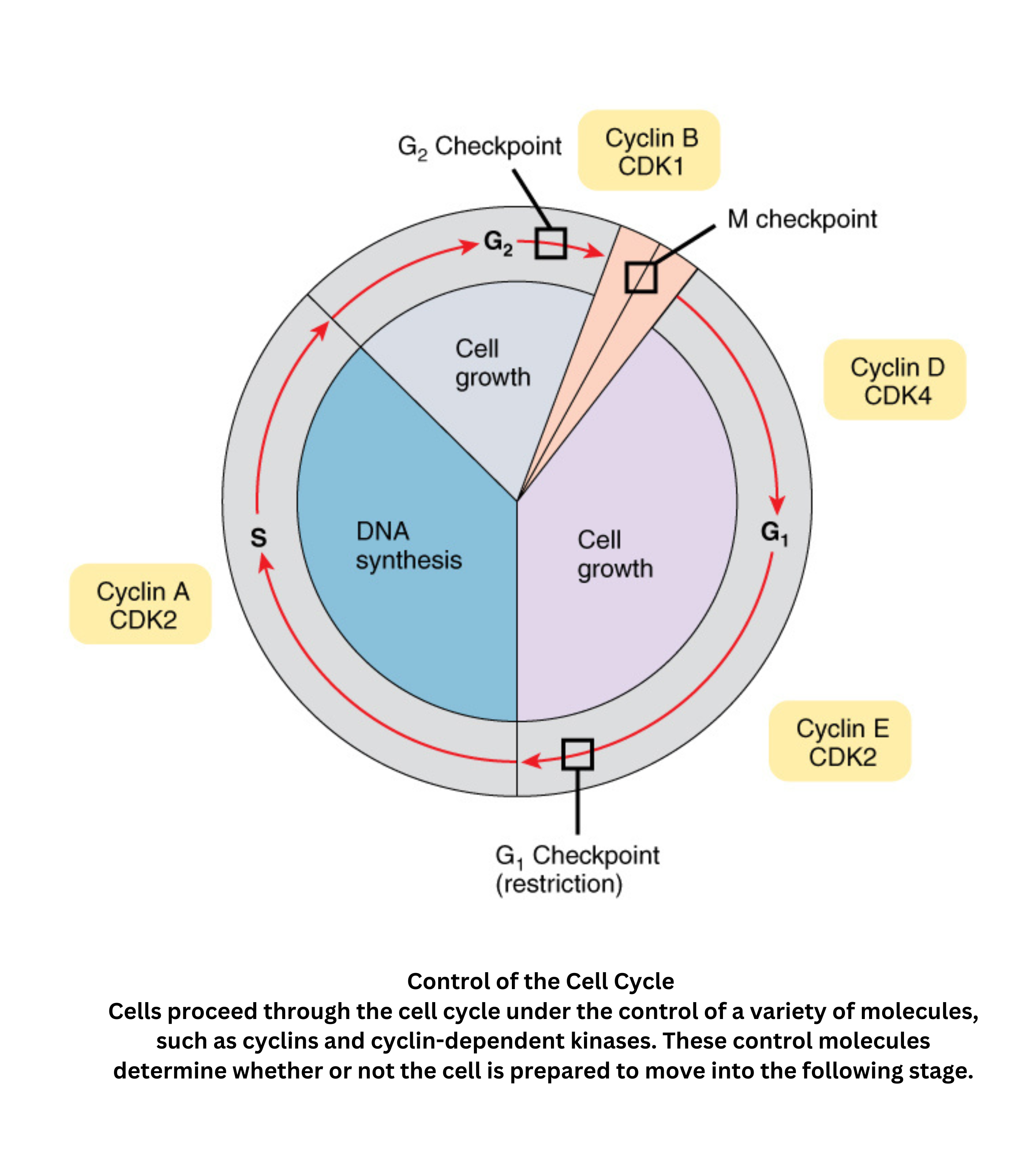
Control of cell cycle
Control of the cell cycle in eukaryotic cells is governed by a network of cyclins and cyclin-dependent kinases (CDKs). These proteins form complexes that drive the cell through its distinct phases: (growth), (DNA synthesis), (further growth and preparation), and (mitosis).
Critical checkpoints, such as the / and / transitions, assess DNA integrity and cellular readiness. Tumor suppressor proteins like p53 and retinoblastoma protein (Rb) help regulate these checkpoints by inducing cell cycle arrest or apoptosis when DNA damage or other cellular stresses are present.
Signaling pathways including PI3K/AKT and MAPK modulate the activity of these regulatory molecules. This integrates external growth signals with internal cell cycle progression, ensuring that division occurs only when conditions are optimal.
Loss of cell cycle controls in cancer cells
Cancer cells often lose normal cell cycle control due to disruptions in regulatory mechanisms. Mutations in genes coding for cyclins, CDKs, or tumor suppressor proteins such as p53 and Rb can allow cells to bypass checkpoints, resulting in unchecked and rapid cell division. This deregulation, combined with abnormal activation of signaling pathways like PI3K/AKT and MAPK, contributes to genomic instability and the accumulation of additional mutations.
As a result, cancer cells divide uncontrollably, leading to tumor formation and metastasis. This failure in regulation can also hamper immune recognition and elimination of abnormal cells, worsening cancer progression. These disruptions - hallmarks of cancer - have motivated targeted therapies, including CDK inhibitors and drugs that restore p53 function, which aim to reestablish proper cell cycle control and limit tumor growth.
Biosignaling
Biosignaling involves two critical types of genes that regulate cell proliferation:
- Oncogenes are genes that drive cell division, and when they become dysregulated, they can lead to cancer (for example, RAS and MYC).
- In contrast, tumor suppressor genes act as a brake on cell proliferation and promote apoptosis; when these genes malfunction, such as with p53 and Rb, cancer can develop.