Core information
Look-alike/sound-alike (LASA) medications
Look-alike, sound-alike (LASA) medications are drugs that can be confused with each other because they:
- Look similar (packaging, color, size, or shape)
- Sound similar (names that are pronounced similarly)
- Have similar doses or strengths
These mix-ups can cause errors during ordering, prescribing, filling, dispensing, or use. Errors caused by these similarities are called LASA errors, and they’re a type of medication error.
Examples of sound-alike medications include Oxynorm and Oxycontin, and bupropion and buspirone.
Confusion can also happen between different strengths of the same medication. For example, warfarin tablets come in strengths from 1 to 10 mg. If the strength is misread or miscommunicated, the patient may be overdosed or underdosed, which can lead to serious (and potentially fatal) adverse effects.
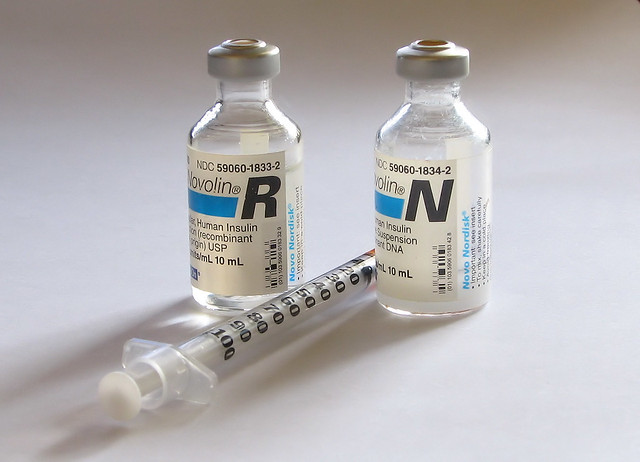
Another common source of LASA errors is mixing up different forms of insulin, such as:
- HumuLIN® R U-500 (Regular insulin human)
- HumuLIN® N NPH (human insulin isophane)
Similar packaging, labeling, and coloring can also cause errors when dispensing or taking look-alike drugs. In previously reported LASA errors, vials of tranexamic acid (used to control bleeding) were confused with bupivacaine or ropivacaine because the bottles were similar in size and had blue caps. These errors are especially common with anesthetic drugs.
LASA errors can have severe outcomes, including overdosing, underdosing, or unintended use of a medication. The risk is especially serious in:
- Pregnant patients
- Older adults
- Very young patients
- Patients with organ dysfunction
Other factors that increase LASA risk include illegible handwriting and having multiple dosage forms (for example, slow-release vs normal release).
The following steps can be taken to prevent LASA errors -
- Use of “tall man” lettering on labels and packaging of error-prone medications, e.g., buPROPion, busPIRone, metFORMIN, and metoPROLOL
- Use color coding for different strengths of the same medication, e.g., Warfarin 2.5 mg tablet is green while Warfarin 4 mg tablet is blue.
- Prescriptions should be written and include details like indication, diagnosis, dose, strength, route of administration, and formulation so that the pharmacist can easily verify them.
- Store LASA medications in separate locations or non-alphabetically. Label bins or shelves with brand names for LASA medications. Store medications so they are easily recognized.
- Keep medication dispensing areas clean and organized.
- Have a double-check system in place to help prevent accidental errors.
- Patient awareness and education about LASA medications.
Medication and dispensing errors and error prevention strategies
Medication errors: Medication errors are any preventable events that may cause or lead to inappropriate medication use or patient harm while the medication is controlled by a healthcare professional, patient, or consumer. Medication errors can occur at any stage, including ordering, prescribing, transcribing, dispensing, administering, or monitoring a drug.
Dispensing errors: Dispensing errors are medication errors linked to the pharmacy (or to the healthcare professional who dispenses the medication). A dispensing error causes a deviation from the prescription and can be classified as follows -
- Dose/item error - when the wrong drug or strength is selected
- Labeling error - incorrect labeling
- Documentation error - correct medication is dispensed, but there are errors in the documentation.
| Type | Example |
|---|---|
| Trailing zero | 1.0 mg read as 10 mg |
| Decimal point error | 0.5 mg read as 5 mg |
| Abbreviation errors | AZT (zidovudine) mistaken as azathioprine |
| Sig abbreviations | Misinterpreting “qod” to “qd” or “AD” to “OD” |
Common reasons for medication and dispensing errors include:
- Failure to communicate drug orders
- Illegible handwriting
- Distractions
- Wrong drug selection from a drop-down menu
- Confusion over similarly named drugs
- Confusion due to similar packaging between products
- Compounding errors
- Errors involving dosing units or weight
Most prescription errors are caused by illegible handwriting, abnormal doses, early refills, incorrect quantity, incorrect patient, or incorrect drug.
Steps to prevent medication and dispensing errors are as follows:
- Refer to detailed patient information such as using multiple patient identifiers (name, year of birth, address, etc.), allergy history, knowledge of current medications to prevent interactions, etc.
- All staff members who prescribe, dispense, administer, or educate patients on medications should have easy access to current drug information and other decision-support resources.
- Certain high-risk medications require barcodes in hospitalized patients. Patients are made to wear wristbands with barcodes on them. Scanning the barcodes allows healthcare professionals to verify that the right drug, dose, route of administration, patient, and time are selected for drug delivery. Examples of high-alert medications include warfarin, insulin, and antiarrhythmics like amiodarone, lidocaine, heparin, magnesium sulfate, and colchicine.
- Detailed and precise prescription information.
- Identify high-risk or high-alert medications. Medication errors can cause serious adverse events, including death.
- Proper storage and labeling of medications.
- Patient education and awareness.
- Prevention of LASA errors (refer to above).
NDC number and UPC code
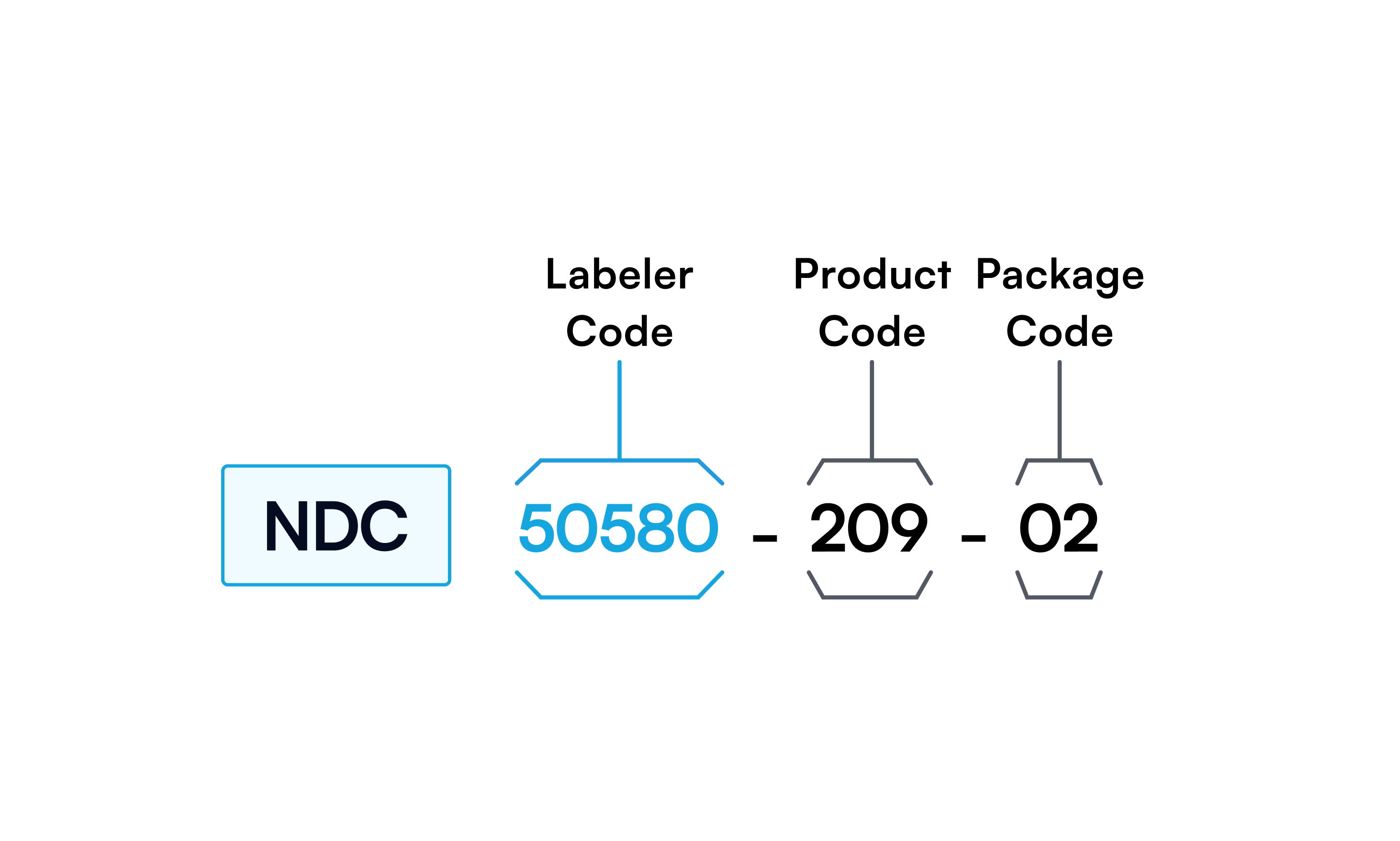
NDC stands for National Drug Code, a unique 10-digit number assigned to prescription and over-the-counter drugs. It helps pharmacy professionals, healthcare providers, and insurance companies identify drugs.
The 10-digit NDC appears in one of the following configurations:
- 4-4-2
- 5-3-2
- 5-4-1
The NDC has three segments:
- Labeler code (first segment, assigned by the FDA)
- Product code (second segment)
- Package code (third segment)
The product code indicates the drug’s strength, form, and formulation. The package code indicates the package form and size.
The NDC helps choose the correct drug and strength. It can also help identify drug-drug interactions, reduce errors, and facilitate processing drug returns.
Generally, the NDC number on the package is a 10-digit number and must be expanded to 11 digits by inserting a “0” in the appropriate spot for insurance and billing purposes. The NDC can be converted into a scannable barcode, known as a UPC code. The UPC code is a 12-digit number.
Event reporting
Like adverse events, medication errors must be reported to regulatory agencies, including the FDA and state pharmacy boards. Reports can be made by healthcare professionals and by patients. These organizations review error submissions collectively, and case reports may be published to educate healthcare professionals about errors and near misses.
In some cases, the FDA may work with drug manufacturers and others to address concerns with pharmaceutical labeling, packaging, and nomenclature and to make changes that reduce the risk of medication errors.
FDA MedWatch: Patients and healthcare practitioners can use FDA MedWatch to report medication errors and vaccine and drug adverse events. The FDA MedWatch reporting system provides a comprehensive sentry position for many medication errors. All errors need to be reported, whether they caused harm to the patient or not.
FAERS: The FDA Adverse Event Reporting System (FAERS) is a database of adverse events and medication errors reported to the FDA. Healthcare professionals, consumers, and manufacturers can submit reports to FAERS.
VAERS: The vaccine adverse event reporting system, or VAERS, is used to report vaccine-related adverse events. It is co-administered by the FDA and CDC.
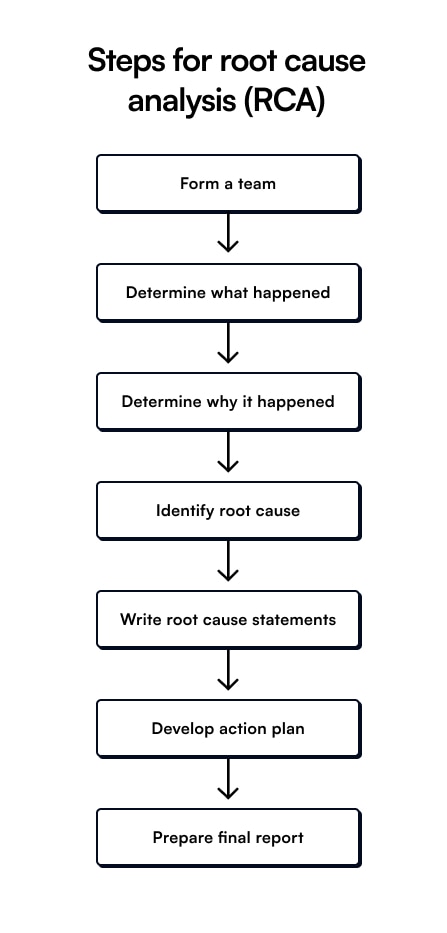
Root-cause analysis (RCA) Root cause analysis consists of procedures used to identify the cause of errors (such as medication errors) and to suggest preventive measures. It is a regulatory requirement and is essential for all sentinel events.
A sentinel event can potentially cause serious harm to the patient and results from systematic errors. For example, a look-alike medication may be dispensed incorrectly due to a chain of problems involving prescribing, inefficient storage, quality control, and human error.